- Biochemistry
-
For the journal, see Biochemistry (journal)."Biological Chemistry" redirects here. For the journal formerly named Biological Chemistry Hoppe-Seyler, see Biological Chemistry (journal).
Biochemistry, sometimes called biological chemistry, is the study of chemical processes in living organisms, including, but not limited to, living matter. Biochemistry governs all living organisms and living processes. By controlling information flow through biochemical signalling and the flow of chemical energy through metabolism, biochemical processes give rise to the incredible complexity of life. Much of biochemistry deals with the structures and functions of cellular components such as proteins, carbohydrates, lipids, nucleic acids and other biomolecules —although increasingly processes rather than individual molecules are the main focus. Over the last 40 years biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine are engaged in biochemical research. Today the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells which in turn relates greatly to the study and understanding of whole organisms.
Among the vast number of different biomolecules, many are complex and large molecules (called biopolymers), which are composed of similar repeating subunits (called monomers). Each class of polymeric biomolecule has a different set of subunit types.[1] For example, a protein is a polymer whose subunits are selected from a set of 20 or more amino acids. Biochemistry studies the chemical properties of important biological molecules, like proteins, and in particular the chemistry of enzyme-catalyzed reactions.
The biochemistry of cell metabolism and the endocrine system has been extensively described. Other areas of biochemistry include the genetic code (DNA, RNA), protein synthesis, cell membrane transport, and signal transduction.
Contents
History
Main article: History of biochemistryIt once was generally believed that life and its materials had some essential property or substance distinct from any found in non-living matter, and it was thought that only living beings could produce the molecules of life.[citation needed] Then, in 1828, Friedrich Wöhler published a paper on the synthesis of urea, proving that organic compounds can be created artificially.[2][3]
The dawn of biochemistry may have been the discovery of the first enzyme, diastase (today called amylase), in 1833 by Anselme Payen. Eduard Buchner contributed the first demonstration of a complex biochemical process outside of a cell in 1896: alcoholic fermentation in cell extracts of yeast. Although the term “biochemistry” seems to have been first used in 1882, it is generally accepted that the formal coinage of biochemistry occurred in 1903 by Carl Neuberg, a German chemist. Previously, this area would have been referred to as physiological chemistry[citation needed]. Since then, biochemistry has advanced, especially since the mid-20th century, with the development of new techniques such as chromatography, X-ray diffraction, dual polarisation interferometry, NMR spectroscopy, radioisotopic labeling, electron microscopy and molecular dynamics simulations. These techniques allowed for the discovery and detailed analysis of many molecules and metabolic pathways of the cell, such as glycolysis and the Krebs cycle (citric acid cycle).
Another significant historic event in biochemistry is the discovery of the gene and its role in the transfer of information in the cell. This part of biochemistry is often called molecular biology. In the 1950s, James D. Watson, Francis Crick, Rosalind Franklin, and Maurice Wilkins were instrumental in solving DNA structure and suggesting its relationship with genetic transfer of information. In 1958, George Beadle and Edward Tatum received the Nobel Prize for work in fungi showing that one gene produces one enzyme. In 1988, Colin Pitchfork was the first person convicted of murder with DNA evidence, which led to growth of forensic science. More recently, Andrew Z. Fire and Craig C. Mello received the 2006 Nobel Prize for discovering the role of RNA interference (RNAi), in the silencing of gene expression.
Starting materials: the chemical elements of life
Main articles: Composition of the human body and Dietary mineralAround two dozen of the 94 naturally-occurring chemical elements are essential to various kinds of biological life. Most rare elements on Earth are not needed by life (exceptions being selenium and iodine), while a few common ones (aluminum and titanium) are not used. Most organisms share element needs, but there are a few differences between plants and animals. For example ocean algae use bromine but land plants and animals seem to need none. All animals require sodium, but some plants do not. Plants need boron and silicon, but animals may not (or may need ultra-small amounts). Just six elements—carbon, hydrogen, nitrogen, oxygen, calcium, and phosphorus—make up almost 99% of the mass of a human body (see composition of the human body for a complete list). In addition to the six major elements that compose most of the human body, humans require smaller amounts of possibly 18 more.[4]
Biomolecules
The four main classes of molecules in biochemistry are carbohydrates, lipids, proteins, and nucleic acids. Many biological molecules are polymers: in this terminology, monomers are relatively small micromolecules that are linked together to create large macromolecules, which are known as polymers. When monomers are linked together to synthesize a biological polymer, they undergo a process called dehydration synthesis.
Carbohydrates
Main articles: Carbohydrates, Monosaccharides, Disaccharides, and PolysaccharidesCarbohydrates are made from monomers called monosaccharides. Some of these monosaccharides include glucose (C6H12O6), fructose (C6H12O6), and deoxyribose (C5H10O4). When two monosaccharides undergo dehydration synthesis, water is produced, as two hydrogen atoms and one oxygen atom are lost from the two monosaccharides' hydroxyl group.
Lipids
Main articles: Lipids, Glycerol, and Fatty acidsLipids are usually made from one molecule of glycerol combined with other molecules. In triglycerides, the main group of bulk lipids, there is one molecule of glycerol and three fatty acids. Fatty acids are considered the monomer in that case, and may be saturated (no double bonds in the carbon chain) or unsaturated (one or more double bonds in the carbon chain).
Lipids, especially phospholipids, are also used in various pharmaceutical products, either as co-solubilisers (e.g. in parenteral infusions) or else as drug carrier components (e.g. in a liposome or transfersome).
Proteins
Main articles: Proteins and Amino Acids The general structure of an α-amino acid, with the amino group on the left and the carboxyl group on the right.
The general structure of an α-amino acid, with the amino group on the left and the carboxyl group on the right.
Proteins are very large molecules – macro-biopolymers – made from monomers called amino acids. There are 20 standard amino acids, each containing a carboxyl group, an amino group, and a side chain (known as an "R" group). The "R" group is what makes each amino acid different, and the properties of the side chains greatly influence the overall three-dimensional conformation of a protein. When amino acids combine, they form a special bond called a peptide bond through dehydration synthesis, and become a polypeptide, or protein.
To determine if two proteins are related or in other words to decide whether they are homologous or not, scientists use sequence-comparison methods. Methods like Sequence Alignments and Structural Alignments are powerful tools that help scientist identify homologies between related molecules.
The relevance of finding homologies among proteins goes beyond forming an evolutionary pattern of protein families. By finding how similar two protein sequences are, we acquire knowledge about their structure and therefore their function.
Nucleic acids
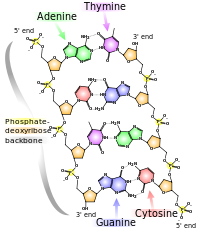
Nucleic acids are the molecules that make up DNA, an extremely important substance which all cellular organisms use to store their genetic information. The most common nucleic acids are deoxyribonucleic acid and ribonucleic acid. Their monomers are called nucleotides. The most common nucleotides are adenine, cytosine, guanine, thymine, and uracil. Adenine binds with thymine and uracil; Thymine only binds with adenine; and cytosine and guanine can only bind with each other.
Carbohydrates
Main article: CarbohydrateThe function of carbohydrates includes energy storage and providing structure. Sugars are carbohydrates, but not all carbohydrates are sugars. There are more carbohydrates on Earth than any other known type of biomolecule; they are used to store energy and genetic information, as well as play important roles in cell to cell interactions and communications.
Monosaccharides
The simplest type of carbohydrate is a monosaccharide, which among other properties contains carbon, hydrogen, and oxygen, mostly in a ratio of 1:2:1 (generalized formula CnH2nOn, where n is at least 3). Glucose, one of the most important carbohydrates, is an example of a monosaccharide. So is fructose, the sugar commonly associated with the sweet taste of fruits.[5][a] Some carbohydrates (especially after condensation to oligo- and polysaccharides) contain less carbon relative to H and O, which still are present in 2:1 (H:O) ratio. Monosaccharides can be grouped into aldoses (having an aldehyde group at the end of the chain, e. g. glucose) and ketoses (having a keto group in their chain; e. g. fructose). Both aldoses and ketoses occur in an equilibrium (starting with chain lengths of C4) cyclic forms. These are generated by bond formation between one of the hydroxyl groups of the sugar chain with the carbon of the aldehyde or keto group to form a hemiacetal bond. This leads to saturated five-membered (in furanoses) or six-membered (in pyranoses) heterocyclic rings containing one O as heteroatom.
Disaccharides
 Sucrose: ordinary table sugar and probably the most familiar carbohydrate.
Sucrose: ordinary table sugar and probably the most familiar carbohydrate.
Two monosaccharides can be joined together using dehydration synthesis, in which a hydrogen atom is removed from the end of one molecule and a hydroxyl group (—OH) is removed from the other; the remaining residues are then attached at the sites from which the atoms were removed. The H—OH or H2O is then released as a molecule of water, hence the term dehydration. The new molecule, consisting of two monosaccharides, is called a disaccharide and is conjoined together by a glycosidic or ether bond. The reverse reaction can also occur, using a molecule of water to split up a disaccharide and break the glycosidic bond; this is termed hydrolysis. The most well-known disaccharide is sucrose, ordinary sugar (in scientific contexts, called table sugar or cane sugar to differentiate it from other sugars). Sucrose consists of a glucose molecule and a fructose molecule joined together. Another important disaccharide is lactose, consisting of a glucose molecule and a galactose molecule. As most humans age, the production of lactase, the enzyme that hydrolyzes lactose back into glucose and galactose, typically decreases. This results in lactase deficiency, also called lactose intolerance.
Sugar polymers are characterised by having reducing or non-reducing ends. A reducing end of a carbohydrate is a carbon atom which can be in equilibrium with the open-chain aldehyde or keto form. If the joining of monomers takes place at such a carbon atom, the free hydroxy group of the pyranose or furanose form is exchanged with an OH-side chain of another sugar, yielding a full acetal. This prevents opening of the chain to the aldehyde or keto form and renders the modified residue non-reducing. Lactose contains a reducing end at its glucose moiety, whereas the galactose moiety form a full acetal with the C4-OH group of glucose. Saccharose does not have a reducing end because of full acetal formation between the aldehyde carbon of glucose (C1) and the keto carbon of fructose (C2).
Oligosaccharides and polysaccharides
 Cellulose as polymer of β-D-glucose
Cellulose as polymer of β-D-glucose
When a few (around three to six) monosaccharides are joined together, it is called an oligosaccharide (oligo- meaning "few"). These molecules tend to be used as markers and signals, as well as having some other uses. Many monosaccharides joined together make a polysaccharide. They can be joined together in one long linear chain, or they may be branched. Two of the most common polysaccharides are cellulose and glycogen, both consisting of repeating glucose monomers.
- Cellulose is made by plants and is an important structural component of their cell walls. Humans can neither manufacture nor digest it.
- Glycogen, on the other hand, is an animal carbohydrate; humans and other animals use it as a form of energy storage.
Use of carbohydrates as an energy source
Main article: Carbohydrate metabolismGlucose is the major energy source in most life forms. For instance, polysaccharides are broken down into their monomers (glycogen phosphorylase removes glucose residues from glycogen). Disaccharides like lactose or sucrose are cleaved into their two component monosaccharides.
Glycolysis (anaerobic)
Glucose is mainly metabolized by a very important ten-step pathway called glycolysis, the net result of which is to break down one molecule of glucose into two molecules of pyruvate; this also produces a net two molecules of ATP, the energy currency of cells, along with two reducing equivalents in the form of converting NAD+ to NADH. This does not require oxygen; if no oxygen is available (or the cell cannot use oxygen), the NAD is restored by converting the pyruvate to lactate (lactic acid) (e. g. in humans) or to ethanol plus carbon dioxide (e. g. in yeast). Other monosaccharides like galactose and fructose can be converted into intermediates of the glycolytic pathway.
Aerobic
In aerobic cells with sufficient oxygen, like most human cells, the pyruvate is further metabolized. It is irreversibly converted to acetyl-CoA, giving off one carbon atom as the waste product carbon dioxide, generating another reducing equivalent as NADH. The two molecules acetyl-CoA (from one molecule of glucose) then enter the citric acid cycle, producing two more molecules of ATP, six more NADH molecules and two reduced (ubi)quinones (via FADH2 as enzyme-bound cofactor), and releasing the remaining carbon atoms as carbon dioxide. The produced NADH and quinol molecules then feed into the enzyme complexes of the respiratory chain, an electron transport system transferring the electrons ultimately to oxygen and conserving the released energy in the form of a proton gradient over a membrane (inner mitochondrial membrane in eukaryotes). Thereby, oxygen is reduced to water and the original electron acceptors NAD+ and quinone are regenerated. This is why humans breathe in oxygen and breathe out carbon dioxide. The energy released from transferring the electrons from high-energy states in NADH and quinol is conserved first as proton gradient and converted to ATP via ATP synthase. This generates an additional 28 molecules of ATP (24 from the 8 NADH + 4 from the 2 quinols), totaling to 32 molecules of ATP conserved per degraded glucose (two from glycolysis + two from the citrate cycle). It is clear that using oxygen to completely oxidize glucose provides an organism with far more energy than any oxygen-independent metabolic feature, and this is thought to be the reason why complex life appeared only after Earth's atmosphere accumulated large amounts of oxygen.
Gluconeogenesis
Main article: GluconeogenesisIn vertebrates, vigorously contracting skeletal muscles (during weightlifting or sprinting, for example) do not receive enough oxygen to meet the energy demand, and so they shift to anaerobic metabolism, converting glucose to lactate. The liver regenerates the glucose, using a process called gluconeogenesis. This process is not quite the opposite of glycolysis, and actually requires three times the amount of energy gained from glycolysis (six molecules of ATP are used, compared to the two gained in glycolysis). Analogous to the above reactions, the glucose produced can then undergo glycolysis in tissues that need energy, be stored as glycogen (or starch in plants), or be converted to other monosaccharides or joined into di- or oligosaccharides. The combined pathways of glycolysis during exercise, lactate's crossing via the bloodstream to the liver, subsequent gluconeogenesis and release of glucose into the bloodstream is called the Cori cycle.[citation needed]
Proteins
Main article: Protein A schematic of hemoglobin. The red and blue ribbons represent the protein globin; the green structures are the heme groups.
A schematic of hemoglobin. The red and blue ribbons represent the protein globin; the green structures are the heme groups.
Like carbohydrates, some proteins perform largely structural roles. For instance, movements of the proteins actin and myosin ultimately are responsible for the contraction of skeletal muscle. One property many proteins have is that they specifically bind to a certain molecule or class of molecules—they may be extremely selective in what they bind. Antibodies are an example of proteins that attach to one specific type of molecule. In fact, the enzyme-linked immunosorbent assay (ELISA), which uses antibodies, is currently one of the most sensitive tests modern medicine uses to detect various biomolecules. Probably the most important proteins, however, are the enzymes. These molecules recognize specific reactant molecules called substrates; they then catalyze the reaction between them. By lowering the activation energy, the enzyme speeds up that reaction by a rate of 1011 or more: a reaction that would normally take over 3,000 years to complete spontaneously might take less than a second with an enzyme. The enzyme itself is not used up in the process, and is free to catalyze the same reaction with a new set of substrates. Using various modifiers, the activity of the enzyme can be regulated, enabling control of the biochemistry of the cell as a whole.
In essence, proteins are chains of amino acids. An amino acid consists of a carbon atom bound to four groups. One is an amino group, —NH2, and one is a carboxylic acid group, —COOH (although these exist as —NH3+ and —COO− under physiologic conditions). The third is a simple hydrogen atom. The fourth is commonly denoted "—R" and is different for each amino acid. There are twenty standard amino acids. Some of these have functions by themselves or in a modified form; for instance, glutamate functions as an important neurotransmitter.
Amino acids can be joined together via a peptide bond. In this dehydration synthesis, a water molecule is removed and the peptide bond connects the nitrogen of one amino acid's amino group to the carbon of the other's carboxylic acid group. The resulting molecule is called a dipeptide, and short stretches of amino acids (usually, fewer than thirty) are called peptides or polypeptides. Longer stretches merit the title proteins. As an example, the important blood serum protein albumin contains 585 amino acid residues.
The structure of proteins is traditionally described in a hierarchy of four levels. The primary structure of a protein simply consists of its linear sequence of amino acids; for instance, "alanine-glycine-tryptophan-serine-glutamate-asparagine-glycine-lysine-…". Secondary structure is concerned with local morphology (morphology being the study of structure). Some combinations of amino acids will tend to curl up in a coil called an α-helix or into a sheet called a β-sheet; some α-helixes can be seen in the hemoglobin schematic above. Tertiary structure is the entire three-dimensional shape of the protein. This shape is determined by the sequence of amino acids. In fact, a single change can change the entire structure. The alpha chain of hemoglobin contains 146 amino acid residues; substitution of the glutamate residue at position 6 with a valine residue changes the behavior of hemoglobin so much that it results in sickle-cell disease. Finally quaternary structure is concerned with the structure of a protein with multiple peptide subunits, like hemoglobin with its four subunits. Not all proteins have more than one subunit.
Ingested proteins are usually broken up into single amino acids or dipeptides in the small intestine, and then absorbed. They can then be joined together to make new proteins. Intermediate products of glycolysis, the citric acid cycle, and the pentose phosphate pathway can be used to make all twenty amino acids, and most bacteria and plants possess all the necessary enzymes to synthesize them. Humans and other mammals, however, can only synthesize half of them. They cannot synthesize isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine. These are the essential amino acids, since it is essential to ingest them. Mammals do possess the enzymes to synthesize alanine, asparagine, aspartate, cysteine, glutamate, glutamine, glycine, proline, serine, and tyrosine, the nonessential amino acids. While they can synthesize arginine and histidine, they cannot produce it in sufficient amounts for young, growing animals, and so these are often considered essential amino acids.
If the amino group is removed from an amino acid, it leaves behind a carbon skeleton called an α-keto acid. Enzymes called transaminases can easily transfer the amino group from one amino acid (making it an α-keto acid) to another α-keto acid (making it an amino acid). This is important in the biosynthesis of amino acids, as for many of the pathways, intermediates from other biochemical pathways are converted to the α-keto acid skeleton, and then an amino group is added, often via transamination. The amino acids may then be linked together to make a protein.
A similar process is used to break down proteins. It is first hydrolyzed into its component amino acids. Free ammonia (NH3), existing as the ammonium ion (NH4+) in blood, is toxic to life forms. A suitable method for excreting it must therefore exist. Different strategies have evolved in different animals, depending on the animals' needs. Unicellular organisms, of course, simply release the ammonia into the environment. Similarly, bony fish can release the ammonia into the water where it is quickly diluted. In general, mammals convert the ammonia into urea, via the urea cycle.
Lipids
Main article: LipidThe term lipid comprises a diverse range of molecules and to some extent is a catchall for relatively water-insoluble or nonpolar compounds of biological origin, including waxes, fatty acids, fatty-acid derived phospholipids, sphingolipids, glycolipids and terpenoids (e.g. retinoids and steroids). Some lipids are linear aliphatic molecules, while others have ring structures. Some are aromatic, while others are not. Some are flexible, while others are rigid.
Most lipids have some polar character in addition to being largely nonpolar. Generally, the bulk of their structure is nonpolar or hydrophobic ("water-fearing"), meaning that it does not interact well with polar solvents like water. Another part of their structure is polar or hydrophilic ("water-loving") and will tend to associate with polar solvents like water. This makes them amphiphilic molecules (having both hydrophobic and hydrophilic portions). In the case of cholesterol, the polar group is a mere -OH (hydroxyl or alcohol). In the case of phospholipids, the polar groups are considerably larger and more polar, as described below.
Lipids are an integral part of our daily diet. Most oils and milk products that we use for cooking and eating like butter, cheese, ghee etc., are composed of fats. Vegetable oils are rich in various polyunsaturated fatty acids (PUFA). Lipid-containing foods undergo digestion within the body and are broken into fatty acids and glycerol, which are the final degradation products of fats and lipids.
Nucleic acids
Main article: Nucleic acidA nucleic acid is a complex, high-molecular-weight biochemical macromolecule composed of nucleotide chains that convey genetic information. The most common nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleic acids are found in all living cells and viruses. Aside from the genetic material of the cell, nucleic acids often play a role as second messengers, as well as forming the base molecule for adenosine triphosphate, the primary energy-carrier molecule found in all living organisms.
Nucleic acid, so called because of its prevalence in cellular nuclei, is the generic name of the family of biopolymers. The monomers are called nucleotides, and each consists of three components: a nitrogenous heterocyclic base (either a purine or a pyrimidine), a pentose sugar, and a phosphate group. Different nucleic acid types differ in the specific sugar found in their chain (e.g. DNA or deoxyribonucleic acid contains 2-deoxyriboses). Also, the nitrogenous bases possible in the two nucleic acids are different: adenine, cytosine, and guanine occur in both RNA and DNA, while thymine occurs only in DNA and uracil occurs in RNA.
Relationship to other "molecular-scale" biological sciences
Researchers in biochemistry use specific techniques native to biochemistry, but increasingly combine these with techniques and ideas from genetics, molecular biology and biophysics. There has never been a hard-line between these disciplines in terms of content and technique. Today the terms molecular biology and biochemistry are nearly interchangeable. The following figure is a schematic that depicts one possible view of the relationship between the fields:
- Biochemistry is the study of the chemical substances and vital processes occurring in living organisms. Biochemists focus heavily on the role, function, and structure of biomolecules. The study of the chemistry behind biological processes and the synthesis of biologically active molecules are examples of biochemistry.
- Genetics is the study of the effect of genetic differences on organisms. Often this can be inferred by the absence of a normal component (e.g. one gene). The study of "mutants" – organisms with a changed gene that leads to the organism being different with respect to the so-called "wild type" or normal phenotype. Genetic interactions (epistasis) can often confound simple interpretations of such "knock-out" or "knock-in" studies.
- Molecular biology is the study of molecular underpinnings of the process of replication, transcription and translation of the genetic material. The central dogma of molecular biology where genetic material is transcribed into RNA and then translated into protein, despite being an oversimplified picture of molecular biology, still provides a good starting point for understanding the field. This picture, however, is undergoing revision in light of emerging novel roles for RNA.
- Chemical Biology seeks to develop new tools based on small molecules that allow minimal perturbation of biological systems while providing detailed information about their function. Further, chemical biology employs biological systems to create non-natural hybrids between biomolecules and synthetic devices (for example emptied viral capsids that can deliver gene therapy or drug molecules).
See also
Main article: Outline of biochemistryLists
- List of biochemistry topics
- List of biochemists
- List of biomolecules
- List of geneticists & biochemists
- List of nucleic acid simulation software
- Important publications in biochemistry (chemistry)
Related topics
- Biological psychiatry
- Biophysics
- Carbon chauvinism
- Chemical ecology
- Computational biomodeling
- EC number
- Hypothetical types of biochemistry
- International Union of Biochemistry and Molecular Biology
- Metabolome
- Metabolomics
- Molecular biology
- Molecular medicine
- Plant biochemistry
- Structural biology
- Stoichiometry
- Small molecule
- Veterinary
Notes
a. ^ It should be noted that fructose is not the only sugar found in fruits. Glucose and sucrose are also found in varying quantities in various fruits, and indeed sometimes exceed the fructose present. For example, 32 % of the edible portion of date is glucose, compared with 23.70 % fructose and 8.20 % sucrose. Conversely, peaches contain more sucrose (6.66 %) than they do fructose (0.93 %) or glucose (1.47 %).[6]
References
- ^ Campbell, Neil A.; Brad Williamson; Robin J. Heyden (2006). Biology: Exploring Life. Boston, Massachusetts: Pearson Prentice Hall. ISBN 0-13-250882-6. http://www.phschool.com/el_marketing.html.
- ^ Wöhler, F. (1828). "Ueber künstliche Bildung des Harnstoffs". Ann. Phys. Chem. 12: 253–256.
- ^ Kauffman, G. B. and Chooljian, S.H. (2001). "Friedrich Wöhler (1800–1882), on the Bicentennial of His Birth". The Chemical Educator 6 (2): 121–133. doi:10.1007/s00897010444a.
- ^ Ultratrace minerals. Authors: Nielsen, Forrest H. USDA, ARS Source: Modern nutrition in health and disease / editors, Maurice E. Shils ... et al.. Baltimore : Williams & Wilkins, c1999., p. 283-303. Issue Date: 1999 URI: [1]
- ^ Whiting, G.C (1970). "Sugars". In A.C. Hulme. The Biochemistry of Fruits and their Products. Volume 1. London & New York: Academic Press. pp. 1=31
- ^ Whiting, G.C. (1970), p.5
Further reading
- Hunter, Graeme K. (2000). Vital Forces: The Discovery of the Molecular Basis of Life. San Diego: Academic Press. ISBN 0-12-361810-X. OCLC 191848148 44187710 162129355 191848148 44187710.
External links
- The Virtual Library of Biochemistry and Cell Biology
- Biochemistry, 5th ed. Full text of Berg, Tymoczko, and Stryer, courtesy of NCBI.
- Biochemistry, 2nd ed. Full text of Garrett and Grisham.
- Biochemistry Animation (Narrated Flash animations.)
- SystemsX.ch - The Swiss Initiative in Systems Biology
- Biochemistry Online Resources – Lists of Biochemistry departments, websites, journals, books and reviews, employment opportunities and events.
Branches of Chemistry Physical chemistry Chemical kinetics · Chemical physics · Electrochemistry · Materials science · Photochemistry · Quantum chemistry · Solid-state chemistry · Spectroscopy · Surface chemistry · Thermochemistry
Organic chemistry Biochemistry · Biophysical chemistry · Bioinorganic chemistry · Bioorganic chemistry · Chemical biology · Medicinal chemistry · Organic chemistry · Organometallic chemistry · Pharmacy · Physical organic chemistry · Polymer chemistry ·
Inorganic chemistry Others biochemical families: prot · nucl · carb (glpr, alco, glys) · lipd (fata/i, phld, strd, gllp, eico) · amac/i · ncbs/i · ttpy/i
Categories:- Biochemistry
- Subjects taught in medical school
Wikimedia Foundation. 2010.