- DAPI
-
DAPI 
 2-(4-amidinophenyl)-1H -indole-6-carboxamidine
2-(4-amidinophenyl)-1H -indole-6-carboxamidineIdentifiers CAS number 28718-90-3 PubChem 2954 ChemSpider 2848 
ChEBI CHEBI:51231 
ChEMBL CHEMBL48217 
Jmol-3D images Image 1
Image 2- [N@H]=C(N)c3ccc(c2cc1ccc(cc1n2)C(=[N@H])N)cc3
[H]/N=C(/c1ccc(cc1)c2cc3ccc(cc3[nH]2)/C(=N/[H])/N)\N
Properties Molecular formula C16H15N5 Molar mass 277.324  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references DAPI or 4',6-diamidino-2-phenylindole is a fluorescent stain that binds strongly to A-T rich regions in DNA. It is used extensively in fluorescence microscopy. DAPI can pass through an intact cell membrane therefore it can be used to stain both live and fixed cells, though it passes through the membrane less efficiently in live cells and therefore the effectiveness of the stain is lower.
Contents
History
DAPI was first synthesised in 1971 in the laboratory of Otto Dann as part of a search for drugs to treat trypanosomiasis although it was unsuccessful as a drug. Further investigation indicated it bound strongly to DNA and became more fluorescent when it did so. This led to its use in identifying mitochondrial DNA in ultracentrifugation in 1975, the first recorded use of DAPI as a fluorescent DNA stain.[1]
Strong fluorescence when bound to DNA led to the rapid adoption of DAPI for fluorescent staining of DNA for fluorescence microscopy. Its use for detecting DNA in plant, metazoa and bacteria cells and virus particles was demonstrated in the late 1970s, and quantitative staining of DNA inside cells was demonstrated in 1977. Use of DAPI as a DNA stain for flow cytometry was also demonstrated around this time.[1]
Fluorescence properties
When bound to double-stranded DNA DAPI has an absorption maximum at a wavelength of 358 nm (ultraviolet) and its emission maximum is at 461 nm (blue). Therefore for fluorescence microscopy DAPI is excited with ultraviolet light and is detected through a blue/cyan filter. The emission peak is fairly broad[2] DAPI will also bind to RNA, though it is not as strongly fluorescent. Its emission shifts to around 500 nm when bound to RNA.[3]
DAPI's blue emission is convenient for microscopists who wish to use multiple fluorescent stains in a single sample. There is some fluorescence overlap between DAPI and green-fluorescent molecules like fluorescein and green fluorescent protein (GFP) but the effect of this is small. Use of spectral unmixing can account for this effect if extremely precise image analysis is required.
Outside of analytical fluorescence light microscopy DAPI is also popular for labeling of cell cultures to detect the DNA of contaminating mycoplasma or virus. The labelled mycoplasma or virus particles in the growth medium fluoresce once stained by DAPI making them easy to detect.
Live cells and toxicity
DAPI can be used for both fixed and live cell staining, though the concentration of DAPI needed for live cell staining is generally much higher than for fixed cells.[4] It is labeled non-toxic in its MSDS[5] and though was not shown to have mutagenicity to E. coli,[6] it is labelled as a known mutagen in manufacturer information.[2] As it is a DNA binding compound it is likely to have some low level mutagenic properties and care should be taken in its handling and disposal.
Alternatives
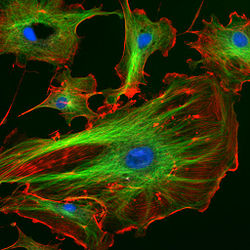 Epithelial cells stained with DAPI (blue) and two antibodies (green and red) via immunofluorescence
Epithelial cells stained with DAPI (blue) and two antibodies (green and red) via immunofluorescence
The Hoechst stains are similar to DAPI in that they are also blue-fluorescent DNA stains which are compatible with both live- and fixed-cell applications.
See also
- Lexitropsin
- Netropsin
- Pentamidine
References
- ^ a b Kapuscinski J (September 1995). "DAPI: a DNA-specific fluorescent probe". Biotech Histochem 70 (5): 220–33. PMID 8580206.
- ^ a b Invitrogen, DAPI Nucleic Acid Stain. accessed 2009-12-08.
- ^ Scott Prahl, DAPI. accessed 2009-12-08.
- ^ Zink D, Sadoni N, Stelzer E. (2003). "Visualizing Chromatin and Chromosomes in Living Cells.". Methods 29 (1): 42–50. doi:10.1016/S1046-2023(02)00289-X. PMID 12543070.
- ^ [1]
- ^ Ohta T, Tokishita S, Yamagata H. (2001). "Ethidium bromide and SYBR Green I enhance the genotoxicity of UV-irradiation and chemical mutagens in E. coli.". Mutat Res. 492 (1-2): 91–7. PMID 11377248.
Categories:- Staining dyes
- Fluorescent dyes
- DNA-binding substances
- Indoles
- Amidines
- [N@H]=C(N)c3ccc(c2cc1ccc(cc1n2)C(=[N@H])N)cc3
Wikimedia Foundation. 2010.

