- Calcium nitrate
-
Calcium nitrate 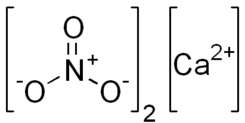 Other namesKalksalpeter, nitrocalcite, Norwegian saltpeter, lime nitrate
Other namesKalksalpeter, nitrocalcite, Norwegian saltpeter, lime nitrateIdentifiers CAS number 10124-37-5  ,
,
13477-34-4 (tetrahydrate)PubChem 24963 ChemSpider 23336 
UN number 1454 RTECS number EW2985000 Jmol-3D images Image 1 - [Ca+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O
Properties Molecular formula Ca(NO3)2 Molar mass 164.088 g/mol (anhydrous)
236.15 (tetrahydrate)Appearance colorless solid Density 2.504 g/cm3 (anhydrous)
1.896 g/cm3 (tetrahydrate)Melting point 561 °C (anhydrous)
42.7 °C (tetrahydrate)Boiling point decomposes (anhydrous)
132 °C (tetrahydrate)Solubility in water anhydrous:
121.2 g/100 mL (20 °C)
271.0 g/100 mL (40° C)
tetrahydrate:
195 g/100 mL (0 °C)
129 g/100 mL (20 °C)
363 g/100 mL (100 °C)Solubility dissolves in alcohol and acetone Structure Crystal structure cubic (anhydrous)
monoclinic (tetrahydrate)Hazards MSDS ICSC 1037 EU Index Not listed NFPA 704 Flash point Non-flammable Related compounds Other anions Calcium sulfate
Calcium chlorideOther cations Magnesium nitrate
Strontium nitrate
Barium nitrate (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Calcium nitrate, also called Norgessalpeter (Norwegian saltpeter) is the inorganic compound with the formula Ca(NO3)2. This colourless salt absorbs moisture from the air and is commonly found as a tetrahydrate. It can also be found in a complex salt as Calcium Ammonium Nitrate decahydrate or Calcium Potassium Nitrate decahydrate. It is mainly used as a component in fertilizers but also technical applications. Nitrocalcite is the name for a mineral which is a hydrated calcium nitrate that forms as an efflorescence where manure contacts concrete or limestone in a dry environment as in stables or caverns.[1]
Contents
Production and reactivity
Norgessalpeter was the first synthetic nitrogen fertilizer compound to be manufactured. Production began at Notodden, Norway in 1905 by the Birkeland–Eyde process. Most of the world's calcium nitrate is now made in Porsgrunn.
It is produced by treating limestone with nitric acid, followed by neutralization with ammonia:
- CaCO3 + 2 HNO3 → Ca(NO3)2 + CO2 + H2O
It is also a byproduct of the Odda Process for the extraction of calcium phosphate, what is the case for the production in Porsgrunn:
- Ca3(PO4)2 + 6 HNO3 + 12 H2O → 2 H3PO4 + 3 Ca(NO3)2 + 12 H2O
It can also be prepared from an aqueous solution of ammonium nitrate, and calcium hydroxide:
- 2 NH4NO3 + Ca(OH)2 → Ca(NO3)2 + 2 NH4OH
Like related alkaline earth metal nitrates (as well as LiNO3), calcium nitrate decomposes upon heating to release nitrogen dioxide:[1]
- 2 Ca(NO3)2 → 2 CaO + 4 NO2 + O2 ΔH = 369 kJ/mol
Use in fertilizer
The fertilizer grade (15.5-0-0 + 19% Ca) is popular in the greenhouse and hydroponics trades; it contains ammonium nitrate and water, as the "double salt" 5Ca(NO3)2.NH4NO3.10H2O. Formulations lacking ammonia are also known: Ca(NO3)2.4H2O (11.9-0-0 + 16.9%Ca). A liquid formulation (9-0-0 + 11% Ca) is also offered. An anhydrous, air-stable derivative is the urea complex Ca(NO3)2.4[OC(NH2)2], which has been sold as Cal-Urea.
Use in technical applications
Calcium Nitrate is used in various technical applications. Most important ones are waste water pre-conditioning for odour emission prevention and set accelerating concrete admixtures.
The waste water pre-conditioning bases on establishing an anoxic biology in the waste water system. At the presence of Nitrate the metabolism for Sulfates is stopped. This results in prevention of hydrogen sulphide formation [2]. Additionally easy degradable organic matter is consumed, what otherwise can cause anaerobic conditions downstream as well as odour emissions itself [3] [4] [5]. The concept is also applicable for surplus sludge treatment [6].
The Calcium Nitrate use within concrete and mortar applications bases on two effects. The Calcium ion leads to an earlier formation of Calcium Hydroxide and thus precipitation sets off faster and the setting time gets reduced [7]. This effect is used also in cold weather concreting agents as well as some combined plasticizers [8] [9] [10]. The Nitrate ion leads to Iron hydroxide formation. Those protective layers reduce the corrosion of the concrete reinforcement [11] [12] [13].
References
- ^ a b Wolfgang Laue, Michael Thiemann, Erich Scheibler, Karl Wilhelm Wiegand “Nitrates and Nitrites” in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim.doi:10.1002/14356007.a17_265. Article Online Posting Date: June 15, 2000
- ^ BENTZEN G, SMITH AT, BENNETT D, WEBSTER NJ, REINHOLD F, SLETHOLT E and HOBSON J (1995): Controlled dosing of nitrate for prevention of H2S in a sewer network and the effects on the subsequent treatment process. Wat. Sci. Tech. Vol 31, No 7, pp. 293-302, 1995.
- ^ ATV-DVWK-M 154 (2003): Geruchsemissionen aus Entwässerungssystemen - Vermeidung oder Verminderung - Ausgabe: Oktober 2003. DWA. ISBN 978-3-924063-82-5.
- ^ FRECHEN FB, FRANKE W (2009): Geruchsemissionen aus Entwässerungsnetzen. DWA Kurs Entwässerungskonzepte N/1, 4.-6.3.2009, Kassel. Proceedings, pp. 349-388, ISBN 978-3-941089-55-6.
- ^ FREY M (2009): Untersuchungen zur Sulfidbildung und zur Effizienz der Geruchsminimierung durch Zugabe von Additiven in Abwasserkanalisationen, Schriftenreihe des Fachgebietes Siedlungswasserwirtschaft der Universitaet Kassel Band 28, Genehmigte Dissertation, ISBN 978-3-89958-453-0, 2009
- ^ EINARSEN AM, ÆESØY A, RASMUSSEN AI, BUNGUM S, SVEBERG M (2000): Biological prevention and removal of hydrogen sulphide in sludge at Lillehammer Wastewater Treatment Plant. Wat. Sci. Tech. Vol 41, No 6, pp. 175-187, 2000.
- ^ JUSTNES, H. and NYGAARD, E.C. (1993): Technical Nitrate as Set Accelerator for Cement. Nordic Concrete Research, Publication No. 13 (1993), pp. 70-87.
- ^ JUSTNES, H. and NYGAARD, E.C. (1995): Technical Calcium Nitrate as Set Accelerator for Cement at Low Temperatures. Cement and Concrete Research, Vol. 25, No. 8, 1995, pp. 1766-1774.
- ^ JUSTNES, H. and PETERSEN, B.G. (2000): Counteracting Retardation of Cement Setting by Plasticizers with Calcium Nitrate. Extended Abstracts of Cement and Concrete Science Conference, Institute of Materials, the University of Sheffield, UK; 11-12 September, 2000, pp. 103-106
- ^ JUSTNES, H. (2010): Calcium Nitrate as a Multifunctional Concrete Admixture. Concrete Magazine, Vol 44, No1, p.34, The concrete Society, UK, 2010. ISSN: 0010-5317
- ^ JUSTNES, H. and NYGAARD, E.C. (1994): The Influence of Technical Calcium Nitrate Additions on the Chloride Binding Capacity of Cement and the Rate of Chloride Induced Corrosion of Steel Embedded in Mortars. Proceedings of the International Conference on Corrosion and Corrosion Protection of Steel in Concrete, Sheffield, UK, July 25-28, 1994, Vol. I, pp. 491-502
- ^ Al-MEHTHEL, M.; Al-DULAIJAN, S.; Al-IDI, S.H. , SHAMEM, M. and ALI, M.R. (2009): Performance of generic and Proprietary corrosion inhibitors in chloride-contaminated silica fume cement concrete. Construction and Building Materials 23 (2009), pp 1768-1774
- ^ Al-AMOUDI, O.S.; MASLEHUDDIN, M.; LASHARI, A.N. and ALMUSSALEM, A.A. (2003): Effectiveness of corrosion inhibitors in contaminated concrete. Cement & Concrete Composites 25(2003), pp. 439-449
External links
Calcium compounds CaB6 · CaBr2 · CaC2 · CaCN2 · CaCO3 · CaC2O4 · CaCl · CaCl2 · Ca(ClO)2 · Ca(ClO3)2 · CaCrO4 · CaF2 · CaH2 · Ca(HCO3)2 · CaH2S2O6 · CaI2 · Ca(IO3)2 · Ca(MnO4)2 · Ca(NO3)2 · CaO · CaO2 · Ca(OH)2 · CaS · CaSO3 · CaSO4 · CaSi2 · CaTiO3 · Ca2P2O7 · Ca2SiO4 · Ca3(AsO4)2 · Ca3(BO3)2 · Ca3(C6H5O7)2 · Ca3N2 · Ca3P2 · Ca3(PO4)2 · Ca(H2PO4)2 · CaHPO4 · C36H70CaO4
Categories:- Calcium compounds
- Nitrates
- Nitrate minerals
- Pyrotechnic oxidizers
- Pyrotechnic colorants
- Fertilizers
- Oxidizing agents
Wikimedia Foundation. 2010.


