- Nitrous acid
-
Nitrous acid  Nitrous acidSystematic nameHydroxidooxidonitrogen
Nitrous acidSystematic nameHydroxidooxidonitrogenIdentifiers CAS number 7782-77-6 
PubChem 24529 
ChemSpider 22936 
EC number 231-963-7 KEGG C00088 
MeSH Nitrous+acid ChEBI CHEBI:25567 
ChEMBL CHEMBL1161681 
Gmelin Reference 983 3DMet B00022 Jmol-3D images Image 1 - O=NO
Properties Molecular formula HNO2 Molar mass 47.013 g/mol Appearance Pale blue solution Density Approx. 1 g/ml Melting point Only known in solution
Acidity (pKa) 3.398 Hazards EU Index Not listed Flash point Non-flammable Related compounds Other anions Nitric acid Other cations Sodium nitrite
Potassium nitrite
Ammonium nitriteRelated compounds Dinitrogen trioxide  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Nitrous acid (molecular formula HNO2) is a weak and monobasic acid known only in solution and in the form of nitrite salts.
Nitrous acid is used to make diazides from amines; this occurs by nucleophilic attack of the amine onto the nitrite, reprotonation by the surrounding solvent, and double-elimination of water. The diazide can then be liberated to give a carbene or carbenoid.
Contents
Structure
In the gas phase, the planar nitrous acid molecule can adopt both a cis and a trans form. The trans form predominates at room temperature, and IR measurements indicate it is more stable by around 2.3 kJ mol−1.[1]
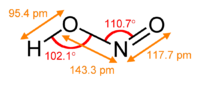
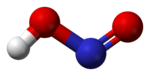
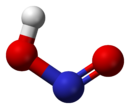
dimensions of the trans form
(from the microwave spectrum)model of the trans form cis form Preparation
When cold, dilute solutions of nitrite ion, NO2− are carefully acidified, a light blue solution of nitrous acid is produced. Free nitrous acid is unstable and decomposes rapidly.
Decomposition
See also: Dinitrogen trioxideIn anything other than very dilute, cold solutions, nitrous acid rapidly decomposes into nitrogen dioxide, nitric oxide, and water:
- 2 HNO2 → NO2 + NO + H2O
Nitrogen dioxide disproportionates into nitric acid and nitrous acid in aqueous solution:[2]
- 2 NO2 + H2O → HNO3 + HNO2
In warm or concentrated solutions, the overall reaction amounts to production of nitric acid, water, and nitric oxide:
- 3 HNO2 → HNO3 + 2 NO + H2O
Chemistry
Nitrous acid is used to prepare diazonium salts:
- HNO2 + ArNH2 + H+ → ArN2+ + 2 H2O
where Ar is an aryl group.
Such salts are widely used in organic synthesis, e.g., for the Sandmeyer reaction and in the preparation azo dyes, brightly-colored compounds that are the basis of a qualitative test for anilines.[3] Nitrous acid is used to destroy toxic and potentially-explosive sodium azide. For most purposes, nitrous acid is usually formed in situ by the action of mineral acid on sodium nitrite:[4]
- NaNO2 + HCl → HNO2 + NaCl
- 2 NaN3 + 2 HNO2 → 3 N2 + 2 NO + 2 NaOH
Reaction with two α-hydrogen atoms in ketones creates oximes, which may be further oxidized to a carboxylic acid, or reduced to form amines. This process is used in the commercial production of adipic acid.
Nitrous acid reacts rapidly with aliphatic alcohols to produce alkyl nitrites, which are potent vasodilators:
- (CH3)2CH-CH2-CH2-OH + HNO2 → (CH3)2CH-CH2-CH2-ONO + H2O
Atmosphere of the earth
Nitrous acid is involved in the ozone budget of the lower atmosphere: the troposphere. The heterogeneous reaction of nitrogen monoxide (NO) and water produces nitrous acid. When this reaction takes place on the surface of atmospheric aerosols, product readily photolyses to hydroxyl radicals.
See also
References
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Oxford: Butterworth-Heinemann. ISBN 0080379419. p. 462
- ^ Kameoka, Yohji; Pigford, Robert (February 1977). "Absorption of Nitrogen Dioxide into Water, Sulfuric Acid, Sodium Hydroxide, and Alkaline Sodium Sulfite Aqueous". Ind. Eng. Chem. Fundamen. 16 (1): 163–169. doi:10.1021/i160061a031.
- ^ Clarke, H. T.; Kirner, W. R. "Methyl Red" Organic Syntheses, Collected Volume 1, p.374 (1941). http://www.orgsyn.org/orgsyn/pdfs/CV1P0374.pdf
- ^ Prudent practices in the laboratory: handling and disposal of chemicals. Washington, D.C.: National Academy Press. 1995. ISBN 0309052297. http://books.nap.edu/openbook.php?record_id=4911&page=165.
Categories:- Nitrogen oxoacids
- Nitrogen metabolism
- Oxidizing agents
Wikimedia Foundation. 2010.
