- Carbamazepine
-
Carbamazepine 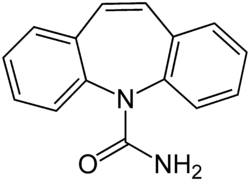
Systematic (IUPAC) name 5H-dibenzo[b,f]azepine-5-carboxamide Clinical data Trade names Tegretol AHFS/Drugs.com monograph MedlinePlus a682237 Pregnancy cat. D(US) Legal status POM (UK) ℞-only (US) Routes Oral Pharmacokinetic data Bioavailability 80% Protein binding 76% Metabolism Hepatic—by CYP3A4, to active epoxide form (carbamazepine-10,11 epoxide) Half-life 25–65 hours (after several doses 12-17 hours) Excretion 2–3% excreted unchanged in urine Identifiers CAS number 298-46-4  85756-57-6
85756-57-6ATC code N03AF01 PubChem CID 2554 DrugBank DB00564 ChemSpider 2457 
UNII 33CM23913M 
KEGG D00252 
ChEBI CHEBI:3387 
ChEMBL CHEMBL108 
Chemical data Formula C15H12N2O Mol. mass 236.269 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Carbamazepine (CBZ) is an anticonvulsant and mood-stabilizing drug used primarily in the treatment of epilepsy and bipolar disorder, as well as trigeminal neuralgia. It is also used off-label for a variety of indications, including attention-deficit hyperactivity disorder (ADHD), schizophrenia, phantom limb syndrome, complex regional pain syndrome, paroxysmal extreme pain disorder, neuromyotonia, intermittent explosive disorder, borderline personality disorder and post-traumatic stress disorder.
It has been seen as safe for pregnant women to use carbamazepine as a mood stabilizer,[1] but, like other anticonvulsants, intrauterine exposure is associated with spina bifida[2] and neurodevelopmental problems.[3]
Contents
Medical uses
Carbamazepine is typically used for the treatment of seizure disorders and neuropathic pain.[4] It may be used as a second line treatment for bipolar disorder and along with antipsychotic agents in schizophrenia.[4]
In the United States, the FDA-approved indications are epilepsy (including partial seizures and tonic-clonic seizures), trigeminal neuralgia, and manic and mixed episodes of bipolar I disorder.[5] Although data are still lacking, carbamazepine appears to be as effective and safe as lithium for the treatment of bipolar disorder, both in the acute and maintenance phase.[6]
Adverse effects
Common adverse effects may include drowsiness, headaches and migraines, motor coordination impairment, and/or upset stomach. Carbamazepine preparations typically greatly decrease a person's alcohol tolerance.
Less common side-effects may include cardiac arrhythmias, blurry or double vision and/or the temporary loss of blood cells or platelets and in rare cases can cause aplastic anemia. With normal use, small reductions in white cell count and serum sodium are common; however, in rare cases, the loss of platelets may become life-threatening. In this case a doctor may recommend frequent blood tests during the first few months of use, followed by three to four tests per year for established patients. Additionally, carbamazepine may possibly exacerbate preexisting cases of hypothyroidism, so yearly thyroid function tests are advisable for persons taking the drug.
There are also rare reports of an auditory side-effect for carbamazepine use, whereby patients perceive sounds about a semitone lower than previously.[7][8][9] Thus, middle C would be heard as the note B3 just below it, and so on. The inverse effect (that is, notes sounding higher) has also been recorded.[10][11] This unusual side-effect is usually not noticed by most people, and quickly disappears after the person stops taking carbamazepine.
Carbamazepine increases the risk of developing lupus by 1.88.[12]
Oxcarbazepine, a derivative of carbamazepine, reportedly has fewer and less serious side-effects.
Carbamazepine may cause Syndrome of inappropriate antidiuretic hormone (SIADH), since it both increases the release and potentiates the action of ADH (vasopressin).
Carbamazepine may aggravate juvenile myoclonic epilepsy, so it is important to uncover any history of jerking, especially in the morning, before starting the drug. It may also aggravate other types of generalized seizure disorder, particularly absence seizures.[13]
In addition, carbamazepine has been linked to serious adverse cognitive anomalies, including EEG slowing[14] and cell apoptosis.[15]
The FDA informed health care professionals that dangerous or even fatal skin reactions (Stevens Johnson syndrome and toxic epidermal necrolysis), that can be caused by carbamazepine therapy, are significantly more common in patients with a particular human leukocyte antigen (HLA) allele, HLA-B*1502. This allele occurs almost exclusively in patients with ancestry across broad areas of Asia, including South Asian Indians.[16] In Europeans a large proportion of sensitivity is associated with HLA-B58.Researchers have also identified another genetic variant, HLA-A*3101 which has been shown to be a strong predictor of both mild and severe adverse reactions to carbamazepine among Japanese[17] and Europeans.[18]
Interactions
Carbamazepine has a potential for drug interactions; caution should be used in combining other medicines with it, including other antiepileptics and mood stabilizers.[5] Lower levels of carbamazepine are seen when administrated with phenobarbital, phenytoin (Dilantin), or primidone (Mysoline). Carbamazepine, as a CYP450 inducer, may increase clearance of many drugs, decreasing their blood levels.[19] Drugs that are more rapidly metabolized with carbamazepine include warfarin (Coumadin), phenytoin (Dilantin), theophylline, and valproic acid (Depakote, Depakote ER, Depakene, Depacon).[5] Drugs that decrease the metabolism of carbamazepine or otherwise increase its levels include erythromycin,[20] cimetidine (Tagamet), propoxyphene (Darvon), and calcium channel blockers.[5] Carbamazepine also increases the metabolism of the hormones in birth control pills and can reduce their effectiveness, potentially leading to unexpected pregnancies.[5]
Valproic acid and valnoctamide both inhibit microsomal epoxide hydrolase (mEH), the enzyme responsible for the breakdown of carbamazepine-10,11 epoxide into inactive metabolites.[21] By inhibiting mEH, valproic acid and valnoctamide cause a buildup of the active metabolite, prolonging the effects of carbamazepine and delaying its excretion.
Grapefruit juice raises the bioavailability of carbamazepine by inhibiting CYP3A4 enzymes in the gut wall and in the liver.
Pharmacokinetics
Carbamazepine exhibits autoinduction: it induces the expression of the hepatic microsomal enzyme system CYP3A4, which metabolizes carbamazepine itself. Upon initiation of carbamazepine therapy, concentrations are predictable and follow their respective baseline clearance/half-life values that have been established for the specific patient. However, after enough carbamazepine has been presented to the liver tissue, the CYP3A4 activity increases, speeding up drug clearance and shortening the half-life. Autoinduction will continue with subsequent increases in dose but will usually reach a plateau within 5–7 days of a maintenance dose. Increases in dose at a rate of 200 mg every 1–2 weeks may be required to achieve a stable seizure threshold. Stable carbamazepine concentrations occur usually within 2–3 weeks after initiation of therapy.[22]
Mechanism of action
The mechanism of action of carbamazepine and its derivatives is relatively well understood. Voltage-gated sodium channels are the molecular pores that allow brain cells (neurons) to generate action potentials, the electrical events that allow neurons to communicate over long distances. After the sodium channels open to start the action potential, they inactivate, in essence closing the channel. Carbamazepine stabilizes the inactivated state of sodium channels, meaning that fewer of these channels are available to subsequently open, making brain cells less excitable (less likely to fire). Carbamazepine has also been shown to potentiate GABA receptors made up of alpha1, beta2, gamma2 subunits.[23]
History
Carbamazepine was discovered by chemist Walter Schindler at J.R. Geigy AG (now part of Novartis) in Basel, Switzerland, in 1953.[24] Schindler then synthesized the drug in 1960, before its anti-epileptic properties had been discovered.
Carbamazepine was first marketed as a drug to treat trigeminal neuralgia (formerly known as tic douloureux) in 1962. It has been used as an anticonvulsant in the UK since 1965, and has been approved in the U.S. since 1974.
In 1971, Drs. Takezaki and Hanaoka first used carbamazepine to control mania in patients refractory to antipsychotics (Lithium was not available in Japan at that time). Dr. Okuma, working independently, did the same thing with success. As they were also epileptologists, they had some familiarity with the anti-aggression effects of this drug. Carbamazepine would be studied for bipolar disorder throughout the 1970s.[25]
Brand names
Carbamazepine has been sold under the names Biston, Calepsin, Carbatrol, Epitol, Equetro, Finlepsin, Sirtal, Stazepine, Telesmin, Tegretol, Teril, Timonil, Trimonil, Epimaz, Carbama/Carbamaze (New Zealand), Amizepin (Poland),Carzine (Kolkata), Karbapin (Serbia), Hermolepsin (Sweden), Degranol (South Africa).,[26] and Tegretal (Chile, Germany).[27]
See also
Chemistry
Schindler, W.; 1960, U.S. Patent 2,948,718.
Carbamazepine is a dibenzazepine.
References
- ^ Gelder, M., Mayou, R. and Geddes, J. 2005. Psychiatry. 3rd ed. New York: Oxford. pp250.
- ^ Jentink, J; Dolk, H, Loane, MA, Morris, JK, Wellesley, D, Garne, E, de Jong-van den Berg, L, EUROCAT Antiepileptic Study Working, Group (2010-12-02). "Intrauterine exposure to carbamazepine and specific congenital malformations: systematic review and case-control study". BMJ (Clinical research ed.) 341: c6581. doi:10.1136/bmj.c6581. PMC 2996546. PMID 21127116. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2996546.
- ^ Cummings, C; Stewart, M, Stevenson, M, Morrow, J, Nelson, J (2011-03-17). "Neurodevelopment of children exposed in utero to lamotrigine, sodium valproate and carbamazepine". Archives of disease in childhood 96 (7): 643–7. doi:10.1136/adc.2009.176990. PMID 21415043.
- ^ a b "Carbamazepine". The American Society of Health-System Pharmacists. http://www.drugs.com/monograph/carbamazepine.html. Retrieved 3 April 2011.
- ^ a b c d e Lexi-Comp (February 2009). "Carbamazepine". The Merck Manual Professional. Archived from the original on 2010-11-18. http://www.merck.com/mmpe/lexicomp/carbamazepine.html. Retrieved on May 3, 2009.
- ^ Ceron-Litvoc D, Soares BG, Geddes J, Litvoc J, de Lima MS (January 2009). "Comparison of carbamazepine and lithium in treatment of bipolar disorder: a systematic review of randomized controlled trials". Hum Psychopharmacol 24 (1): 19–28. doi:10.1002/hup.990. PMID 19053079.
- ^ Yoshikawa H, Abe T (March 2003). "Carbamazepine-induced abnormal pitch perception". Brain Dev. 25 (2): 127–9. doi:10.1016/S0387-7604(02)00155-9. PMID 12581810. http://linkinghub.elsevier.com/retrieve/pii/S0387760402001559.
- ^ Konno S, Yamazaki E, Kudoh M, Abe T, Tohgi H (September 2003). "Half pitch lower sound perception caused by carbamazepine". Intern. Med. 42 (9): 880–3. doi:10.2169/internalmedicine.42.880. PMID 14518681. http://joi.jlc.jst.go.jp/JST.Journalarchive/internalmedicine1992/42.880?from=PubMed.[dead link]
- ^ Kashihara K, Imai K, Shiro Y, Shohmori T (September 1998). "Reversible pitch perception deficit due to carbamazepine". Intern. Med. 37 (9): 774–5. doi:10.2169/internalmedicine.37.774. PMID 9804087. http://joi.jlc.jst.go.jp/JST.Journalarchive/internalmedicine1992/37.774?from=PubMed.[dead link]
- ^ Miyaoka T, Seno H, Itoga M, Horiguchi J (2000). "Reversible pitch perception deficit caused by carbamazepine". Clin Neuropharmacol 23 (4): 219–21. doi:10.1097/00002826-200007000-00010. PMID 11020128. Archived from the original on 2010-11-18. http://meta.wkhealth.com/pt/pt-core/template-journal/lwwgateway/media/landingpage.htm?issn=0362-5664&volume=23&issue=4&spage=219.
- ^ Wakamoto H, Kume A, Nakano N (June 2004). "Elevated pitch perception owing to carbamazepine-activating effect on the peripheral auditory system: auditory brainstem response study". J. Child Neurol. 19 (6): 453–5. PMID 15446396. Archived from the original on 2010-11-18. http://jcn.sagepub.com/cgi/pmidlookup?view=long&pmid=15446396.
- ^ Schoonen, W. Marieke; Thomas, Sara L.; Somers, Emily C.; Smeeth, Liam; Kim, Joseph; Evans, Stephen; Hall, Andrew J. (2010). "Do selected drugs increase the risk of lupus? A matched case-control study". British Journal of Clinical Pharmacology 70 (4): 588–596. doi:10.1111/j.1365-2125.2010.03733.x. PMC 2950993. PMID 20840450. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2950993.
- ^ Lige Liu, Thomas Zheng, Margaret J. Morris, Charlott Wallengren, Alison L. Clarke, Christopher A. Reid, Steven Petrou and Terence J. O'Brien (2006). "The Mechanism of Carbamazepine Aggravation of Absence Seizures". JPET 319 (2): 790–798. doi:10.1124/jpet.106.10496. PMID 16895979. http://jpet.aspetjournals.org/content/319/2/790.abstract.
- ^ Salinsky MC, Binder LM, Oken BS, Storzbach D, Aron CR, Dodrill CB (2002). "Effects of gabapentin and carbamazepine on the EEG and cognition in healthy volunteers". Epilepsia 43 (5): 482–90. doi:10.1046/j.1528-1157.2002.22501.x. PMID 12027908. Archived from the original on 2010-11-18. http://www.blackwell-synergy.com/doi/full/10.1046/j.1528-1157.2002.22501.x?cookieSet=1.
- ^ Gao XM, Margolis RL, Leeds P, Hough C, Post RM, Chuang DM (1995). "Carbamazepine induction of apoptosis in cultured cerebellar neurons: effects of N-methyl-D-aspartate, aurintricarboxylic acid and cycloheximide". Brain Res. 703 (1–2): 63–71. doi:10.1016/0006-8993(95)01066-1. PMID 8719616.
- ^ MedWatch (2007-12-12). "Carbamazepine". 2007 Safety Alerts for Drugs, Biologics, Medical Devices, and Dietary Supplements. FDA. Archived from the original on 2010-11-18. http://www.fda.gov/medwatch/safety/2007/safety07.htm#carbamazepine.
- ^ Ozeki T, Mushiroda T, Yowang A, Takahashi A, Kubo M, Shirakata Y, Ikezawa Z, Iijima M, Shiohara T, Hashimoto K, Kamatani N, Nakamura Y. (March 2011). "Genome-wide association study identifies HLA-A*3101 allele as a genetic risk factor for carbamazepine-induced cutaneous adverse drug reactions in Japanese population". Hum Mol Genet 20 (5): 1034–1041. doi:10.1093/hmg/ddq537. PMID 21149285.
- ^ [1], Epilepsy Society: genome-wide association study of Europeans with adverse reaction to carbamazepine.
- ^ "eMedicine - Toxicity, Carbamazepine". Archived from the original on 2008-08-04. http://www.emedicine.com/emerg/topic77.htm.
- ^ Stafstrom CE, Nohria V, Loganbill H, Nahouraii R, Boustany RM, DeLong GR (January 1995). "Erythromycin-induced carbamazepine toxicity: a continuing problem". Arch Pediatr Adolesc Med 149 (1): 99–101. PMID 7827672. Archived from the original on 2010-11-18. http://archpedi.ama-assn.org/cgi/pmidlookup?view=long&pmid=7827672.
- ^ Gonzalez, Frank J.; Robert H. Tukey (2006). "Drug Metabolism". In Laurence Brunton, John Lazo, Keith Parker (eds.). Goodman & Gilman's The Pharmacological Basis of Therapeutics (11th ed.). New York: McGraw-Hill. p. 79. ISBN 978-0-07-142280-2.
- ^ Bauer, Larry A. (2008). Applied Clinical Pharmacokinetics (2nd ed.). McGraw-Hill. ISBN 978-0-8385-0388-1.
- ^ Granger, P. et al. Modulation of the gamma-aminobutyric acid type A receptor by the antiepileptic drugs carbamazepine and phenytoin. Mol. Pharmacol. 47, 1189–1196 (1995).
- ^ Schindler W, Häfliger F (1954). "Über Derivate des Iminodibenzyls". Helvetica Chimica Acta 37 (2): 472–83.. doi:10.1002/hlca.19540370211.
- ^ Okuma T, Kishimoto A (February 1998). "A history of investigation on the mood stabilizing effect of carbamazepine in Japan". Psychiatry Clin. Neurosci. 52 (1): 3–12. doi:10.1111/j.1440-1819.1998.tb00966.x. PMID 9682927.
- ^ Degranol Tablets[dead link]
- ^ http://www.farmaciasahumada.cl/fasaonline/fasa/MFT/PRODUCTO/P1163.HTM
- ^ Hung SI, Chung WH, Chen YT et al. Common risk allele in aromatic antiepileptic-drug induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Han Chinese. Pharmacogenomics.2010 Mar;11(3):349-56.
- ^ Carbamazepine-Induced Toxic Effects and HLA-B*1502 Screening in Taiwan, New England Journal of Medicine, March 2010. http://www.nejm.org/doi/full/10.1056/NEJMoa1009717
External links
- Carbatrol website
- Equetro website
- Carbamazepine Pharmacokinetics - PubPK
- TA warning
- Carbamazepine overview from PsychEducation.org
- Extensive review of the effects of carbamazepine in pregnancy and breastfeeding (free full text with registration)
- U.S. Patent 2,948,718, August 1960
Anticonvulsants (N03) GABAA receptor agonist Clobazam • Clonazepam • Clorazepate • Diazepam# • Flutoprazepam • Lorazepam • Midazolam • Nimetazepam • Nitrazepam • TemazepamOther GABA agents Carbonic anhydrase inhibitor Channel blockers Primarily sodiumPrimarily calciumUnknown/ungroupedChannel openers PotassiumRetigabineIndirect GABA agents GABA transaminase inhibitor: Valproic acid# (Sodium valproate & Valproate semisodium) • Valpromide • Valnoctamide • Valproate pivoxil
GABA reuptake inhibitor: TiagabineUnknown/multiple/
unsortedPropionatesMood stabilizers Carbamazepine • Divalproex sodium • Gabapentin • Lamotrigine • Licarbazepine • Lithium • Omega-3 fatty acids • Oxcarbazepine • Pregabalin • Sodium valproate • Tiagabine • Topiramate • Valproic acidMood disorder (F30–F39, 296) History Symptoms Spectrum Bipolar disorder (Bipolar I, Bipolar II, Bipolar NOS) · Cyclothymia · Dysthymia · Major depressive disorder · Schizoaffective disorderTreatment Carbamazepine · Gabapentin · Lamotrigine · Oxcarbazepine · Topiramate · Valproic acid (Sodium valproate, Valproate semisodium)Other mood stabilizersNon-pharmaceuticalRelated Categories:- Anticonvulsants
- Mood stabilizers
- Prodrugs
- Ureas
- World Health Organization essential medicines
- Dibenzazepines
Wikimedia Foundation. 2010.

