- Organosulfur compounds
-
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odours, but many of the sweetest compounds known are organosulfur derivatives. Nature abounds with organosulfur compounds—sulfur is essential for life. Two of the 20 common amino acids are organosulfur compounds. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries.
Sulfur shares the chalcogen group with oxygen, and it is expected that organosulfur compounds have similarities with carbon-oxygen compounds, which is true to some extent.
A classical chemical test for the detection of sulfur compounds is the Carius halogen method.
Contents
Classes of organosulfur compounds
Organosulfur compounds can be classified according to the sulfur-containing functional groups, which are listing in decreasing order of their occurrence.
- Illustrative organosulfur compounds
-
Allicin, the active ingredient in garlic
-
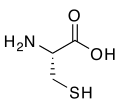
R-cysteine, an amino acid containing a thiol group
-
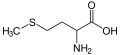
Methionine, an amino acid containing a thioether
-
Diphenyl disulfide, a representative disulfide
-
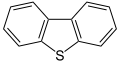
Dibenzothiophene, a component of crude oil
-
Perfluorooctanesulfonic acid, a controversial surfactant
Thioethers, thioesters, thioacetals
These compounds are characterized by C–S–C bonds[1][2] Relative to C–C bonds, C–S bond are both longer, because S is larger than carbon, and about 10% weaker. Representative bond lengths in sulfur compounds are 183 pm for the S-C single bond in methanethiol and 173 pm in thiophene. The C–S bond dissociation energy for thiomethane is 89 kcal/mol (370 kJ/mol) compared to methane's 100 kcal/mol (420 kJ/mol) and when hydrogen is replaced by a methyl group the energy decreases to 73 kcal/mol (305 kJ/mol).[3] The single carbon to oxygen bond is shorter than that of the C–C bond. The bond dissociation energies for dimethyl sulfide and dimethyl ether are respectively 73 and 77 kcal/mol (305 and 322 kJ/mol.
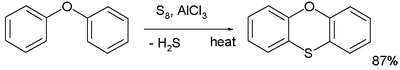
Thioethers are typically prepared by alkylation of thiols. They can also be prepared via the Pummerer rearrangement. In one named reaction called the Ferrario reaction phenyl ether is converted to phenoxathiin by action of elemental sulfur and aluminium chloride [4]
Thioacetals and thioketals feature C–S–C–S–C bond sequence. They represent a subclass of thioethers. The thioacetals are useful in "umpolung" of carbonyl groups.
Thioesters have general structure R–CO–S–R. They are related to regular esters but are more reactive.
Thiophenes represent a special class of thioethers that are aromatic. The resonance stabilization of thiophene is 29 kcal/mol (121 kJ/mol) compared to 20 kcal/mol (84 kJ/mol) for the oxygen analogue furan. The reason for this difference is the higher electronegativity for oxygen drawing away electrons to itself at the expense of the aromatic ring current. Yet as an aromatic substituent the thio group is less effective as an activating group than the alkoxy group.
Thiols, disulfides, polysulfides
Thiol group contain the functionality R–SH. Thiols are structurally similar to the alcohol group, but these functionalities are very different in their chemical properties. Thiols are more nucleophilic, more acidic, and more readily oxidized. This acidity can differ by 5 pKa units.[5]
The difference in electronegativity between sulfur (2.58) and hydrogen (2.20) is small and therefore hydrogen bonding in thiols is not prominent. Aliphatic thiols form monolayers on gold, which are topical in nanotechnology.
Certain aromatic thiols can be accessed through a Herz reaction.
Disulfides R–S–S–R with a covalent sulfur to sulfur bond are important for crosslinking: in biochemistry for the folding and stability of some proteins and in polymer chemistry for the crosslinking of rubber.
Longer sulfur chains are also known, such as in the natural product varacin which contains an unusual pentathiepin ring (5-sulfur chain cyclised onto a benzene ring)
Compounds with double bonds between carbon and sulfur are relatively uncommon, but include important compounds carbon disulfide, carbonyl sulfide, and thiophosgene. Thioketones (RC(=S)R') are uncommon with alkyl substituents, but one example example is thiobenzophenone. Thioaldehydes are rarer still, reflecting their lack of steric protection ("thioformaldehyde" exists as a cyclic trimer). Thioamides, with the formula R1C(=S)N(R2)R3 are more common. They are typically prepared by the reaction of amides with Lawesson's reagent.
Compounds that contain C=S double bonds exist in sulfonium ylides for instance in the Johnson-Corey-Chaykovsky reaction.
Triple bonds between carbon and sulfur
Triple bonds between sulfur and carbon in sulfaalkynes are rare and can be found in carbon monosulfide (CS) [6] and have been suggested for the compounds F3CCSF3 [7][8] and F5SCSF3 [9]. The compound HCSOH is also presented as having a formal triple bond [10].
Thiocarboxylic acids and thioamides
Thiocarboxylic acids (RC(O)SH)) and dithiocarboxylic acids (RC(S)SH) are well known. They are structurally similar to carboxylic acids but more acidic. Thioamides are analogous to amides.
Sulfonic acids, esters, amides
Sulfonic acids have functionality RS(=O)2OH[11]. They are strong acids that are typically soluble in organic solvents. Sulfonic acids like trifluoromethanesulfonic acid is a frequently used reagent in organic chemistry. Sulfa drugs are sulfonamides derived from aromatic sulfonation.
Sulfuranes and persulfuranes
Sulfuranes are relatively specialized functional group that are tetravalent, hypervalent sulfur compounds, with the formula SR4 [12] and likewise persulfuranes are hexavalent SR6. All-carbon persulfuranes have been known for the heavier representatives of the chalcogen group, for instance the compound hexamethylpertellurane (Te(Me)6) was discovered in 1990 [13] by reaction of tetramethyltellurium with xenon difluoride to Te(Me)2)F2 followed by reaction with diethylzinc. The sulfur analogue hexamethylpersulfurane SMe6 has been predicted to be stable [14] but has not been synthesized yet.
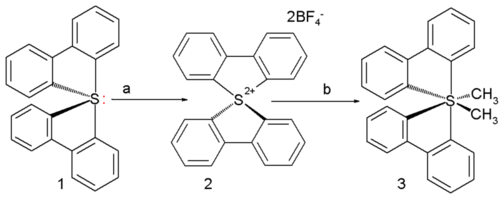
The first ever all-carbon persulfurane actually synthesized in a laboratory has two methyl and two biphenyl ligands [15]:
It is prepared from the corresponding sulfurane 1 with xenon difluoride / boron trifluoride in acetonitrile to the sulfuranyl dication 2 followed by reaction with methyllithium in tetrahydrofuran to (a stable) persulfurane 3 as the cis isomer. X-ray diffraction shows C-S bond lengths ranging between 189 and 193 pm (longer than the standard bond length) with the central sulfur atom in a distorted octahedral molecular geometry.
Computer simulation suggests that these bonds are very polar with the negative charges residing on carbon.
Naturally occurring organosulfur compounds
Not all organosulfur compounds are foul-smelling pollutants. Compounds like allicin and ajoene are responsible for the odor of garlic, and lenthionine contributes to the flavor of shiitake mushrooms. Many of these natural products also have important medicinal properties such as preventing platelet aggregation or fighting cancer.
Organosulfur compounds in pollution
Most organic sulfur compounds in the environment are naturally occurring, as a consequence of the fact that sulfur is essential for life and two amino acids contain this element.
Some organosulfur compounds in the environment, are generated as minor by-products of industrial processes such as the manufacture of plastics and tires.
Selected smell-producing processes are organosulfur compounds produced by the coking of coal designed to drive out sulfurus compounds and other volatile impurities in order to produce 'clean carbon' (coke), which is primarily used for steel production.
Organosulfur compounds in fossil fuels
Odours occur as well in chemical processing of coal or crude oil into precursor chemicals (feedstocks) for downstream industrial uses (e.g. plastics or pharmaceutical production) and the ubiquitous needs of petroleum distillation for (gasolines, diesel, and other grades of fuel oils production.
Organosulfur compounds might be understood as smelly contaminants that need to be removed from natural gas before commercial uses, from exhaust stacks and exhaust vents before discharge. In this latter context, organosulfur compounds may be said to account for the pollutants in sulfurous acid rain, or equivalently, said to be pollutants within most common fossil fuels, especially coal.
See also
CH He CLi CBe CB CC CN CO CF Ne CNa CMg CAl CSi CP CS CCl CAr CK CCa CSc CTi CV CCr CMn CFe CCo CNi CCu CZn CGa CGe CAs CSe CBr CKr CRb CSr CY CZr CNb CMo CTc CRu CRh CPd CAg CCd CIn CSn CSb CTe CI CXe CCs CBa CHf CTa CW CRe COs CIr CPt CAu CHg CTl CPb CBi CPo CAt Rn Fr Ra Rf Db Sg Bh Hs Mt Ds Rg Cn Uut Uuq Uup Uuh Uus Uuo ↓ CLa CCe CPr CNd CPm CSm CEu CGd CTb CDy CHo CEr CTm CYb CLu Ac Th Pa CU Np Pu Am Cm Bk Cf Es Fm Md No Lr Chemical bonds to carbon Core organic chemistry Many uses in chemistry Academic research, but no widespread use Bond unknown / not assessed External links
- Organosulfur chemistry at http://users.ox.ac.uk Link
References
- ^ Organic chemistry IUPAC Blue Book. Rules C-5: Compounds Containing Bivalent Sulfur http://www.acdlabs.com/iupac/nomenclature/79/r79_25.htm
- ^ Organic chemistry IUPAC Blue Book. Recommendation R-5.7.1.3.4 Thiocarboxylic and thiocarbonic acids.http://www.acdlabs.com/iupac/nomenclature/93/r93_480.htm
- ^ Handbook of Chemistry and Physics, 81st Edition CRC Press ISBN 0-8493-0481-4
- ^ Organic Syntheses, Coll. Vol. 2, p.485 (1943); Vol. 18, p.64 (1938). Article link
- ^ R. J. Cremlyn “An Introduction to Organosulfur Chemistry” John Wiley and Sons: Chichester (1996). ISBN 0 471 95512 4.
- ^ Moltzen, E. K.; Klabunde, K. J.; Senning, A. (1988). "Carbon monosulfide: a review". Chemical Reviews 88 (2): 391. doi:10.1021/cr00084a003.
- ^ [Nomenclature Rules C-5: Compounds Containing Bivalent Sulfur]Pötter, B.; Seppelt, K. (1984). "Trifluoroethylidynesulfur Trifluoride, F3C-C≡SF3". Angewandte Chemie International Edition in English 23 (2): 150. doi:10.1002/anie.198401501.
- ^ Buschmann, J.; Damerius, R.; Gerhardt, R.; Lentz, D.; Luger, P.; Marschall, R.; Preugschat, D.; Seppelt, K. et al. (1992). "(Trifluoroethylidyne)sulfur trifluoride, F3CC.tplbond.SF3: two solid-state structures and reactivity as a carbene". Journal of the American Chemical Society 114: 9465. doi:10.1021/ja00050a027.
- ^ Gerhardt, R.; Gerlbig, T.; Buschamann, J. �R.; Luger, P.; Seppelt, K. (1988). "The SF5-Unit as Steric Protecting Group; Synthesis and Structure of F5S–C≡SF3". Angewandte Chemie International Edition in English 27 (11): 1534. doi:10.1002/anie.198815341.
- ^ Schreiner, P.; Reisenauer, H.; Romanski, J.; Mloston, G. (2009). "A formal carbon-sulfur triple bond: H-Ctriple bondS-O-H.". Angewandte Chemie (International ed. in English) 48 (43): 8133–8136. doi:10.1002/anie.200903969. PMID 19768827.
- ^ Organic chemistry IUPAC Blue Book.C-6 Sulfur Halides, Sulfoxides, Sulfones, and Sulfur Acids and Their Derivatives http://www.acdlabs.com/iupac/nomenclature/79/r79_26.htm
- ^ For an example bis[2,2,2-trifluoro-1-phenyl-1-(trifluoromethyl) ethoxy] diphenyl sulfurane Organic Syntheses, Coll. Vol. 6, p.163 (1988); Vol. 57, p.22 (1977) Link.
- ^ Synthesis and characterization of hexamethyltellurium(VI) Latif Ahmed, John A. Morrison J. Am. Chem. Soc.; 1990; 112(20); 7411-7413. Abstract
- ^ The S6 Point Group Conformers of the Hexamethylchalcogens: Me6S, Me6Se, Me6Te Fowler, J. E.; Schaefer, H. F., III; Raymond, K. N. Inorg. Chem.; (Article); 1996; 35(2); 279-281. doi: 10.1021/ic940240d
- ^ Isolation and Molecular Structure of the Organo-persulfuranes [12-S-6(C6)] Sato, S.; Matsunaga, K.; Horn, E.; Furukawa, N.; Nabeshima, T. J. Am. Chem. Soc.; (Communication); 2006; 128(21); 6778-6779. doi:10.1021/ja060497y
Categories:
Wikimedia Foundation. 2010.