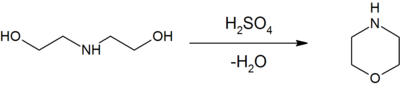- Morpholine
-
Not to be confused with morphine or morpholino.
Morpholine 


 morpholineOther namesDiethylenimide Oxide
morpholineOther namesDiethylenimide Oxide
1,4-oxazinane
tetrahydro-1,4-oxazineIdentifiers CAS number 110-91-8 
UNII 8B2ZCK305O 
KEGG C14452 
ChEMBL CHEMBL276518 
RTECS number QD6475000 Jmol-3D images Image 1 - O(CCNC1)C1
Properties Molecular formula C4H9NO Molar mass 87.1 g/mol Appearance Colorless liquid Density 1.007 g/cm3 Melting point -5 °C, 268 K, 23 °F
Boiling point 129 °C, 402 K, 264 °F
Solubility in water miscible Acidity (pKa) 8.36[1] Hazards MSDS Oxford MSDS R-phrases R10 R20/21/22 R34 S-phrases (S1/2) S23 S36 S45 Main hazards Flammable, Corrosive NFPA 704 Flash point 31 °C Autoignition
temperature275 °C  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Morpholine is an organic chemical compound having the chemical formula O(CH2CH2)2NH. This heterocycle, pictured at right, features both amine and ether functional groups. Because of the amine, morpholine is a base; its conjugate acid is called morpholinium. For example, neutralizing morpholine with hydrochloric acid makes the salt morpholinium chloride.
Contents
Production
Morpholine may be produced by the dehydration of diethanolamine with sulfuric acid:[2]
Uses
Industrial applications
Morpholine is a common additive, in parts per million concentrations, for pH adjustment in both fossil fuel and nuclear power plant steam systems. Morpholine is used because its volatility is about the same as water, so once it is added to the water, its concentration becomes distributed rather evenly in both the water and steam phases. Its pH adjusting qualities then become distributed throughout the steam plant to provide corrosion protection. Morpholine is often used in conjunction with low concentrations of hydrazine or ammonia to provide a comprehensive all-volatile treatment chemistry for corrosion protection for the steam systems of such plants. Morpholine decomposes reasonably slowly in the absence of oxygen at the high temperatures and pressures in these steam systems.
Organic synthesis
Morpholine undergoes most chemical reactions typical for other secondary amines, though the presence of the ether oxygen withdraws electron density from the nitrogen, rendering it less nucleophilic (and less basic) than structurally similar secondary amines such as piperidine. For this reason, it forms a stable chloramine (CAS#23328-69-0).[3]
It is commonly used to generate enamines.[4]
Morpholine is widely used in organic synthesis. For example, it is a building block in the preparation of the antibiotic linezolid and the anticancer agent gefitinib (Iressa).
In research and in industry, the low cost and polarity of morpholine lead to its common use as a solvent for chemical reactions.
Producers
Most (as above) is widespread in Europe and the USA; accordingly, producers in Europe and the USA are able to cover the domestic and export markets.
Agriculture
As a fruit coating
Morpholine is used as a chemical emulsifier in the process of waxing fruit. Naturally, fruits make waxes to protect against insects and fungal contamination, but this can be lost as the fruit is cleaned. A small amount of new wax is applied to replace it. Morpholine is used as an emulsifier and solubility aid for shellac, which is used as a wax for fruit coating.[5]
As a component in fungicides
Morpholine derivatives used as agricultural fungicides in cereals are known as ergosterol biosynthesis inhibitors.
- Amorolfine
- Fenpropimorph
- Tridemorph
References
- ^ Hall, H.K., J. Am. Chem. Soc., 1957, 79, 5441.
- ^ Klaus Weissermel, Hans-Jürgen Arpe, Charlet R. Lindley, Stephen Hawkins (2003). "Chap. 7. Oxidation Products of Ethylene". Industrial Organic Chemistry. Wiley-VCH. pp. 159–161. ISBN 3527305785.
- ^ Lindsay Smith, J. R.; McKeer, L. C.; Taylor, J. M. (1993), "4-Chlorination of Electron-Rich Benzenoid Compounds: 2,4-Dichloromethoxybenzene", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=CV8P0167; Coll. Vol. 8: 167
- ^ Noyori, R.; Yokoyama, K.; Hayakawa, Y. (1988), "Cyclopentenones from α,α'-Dibromoketones and Enamines: 2,5-Dimethyl-3-Phenyl-2-Cyclopenten-1-one", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=CV6P0520; Coll. Vol. 6: 520
- ^ Raymond G. McGuire; Dimitrios A. Dimitroglou,"Evaluation of Shellac and Sucrose Ester Fruit Coating Formulations that Support Biological Control of Post-harvest Grapefruit Decay". Bio-control Science and Technology, Volume 9, Issue 1 March 1999 , pages 53 - 65
Categories:- Morpholines
- Ether solvents
- Amine solvents
Wikimedia Foundation. 2010.