- DLG2
-
Discs, large homolog 2 (Drosophila) 
PDB rendering based on 1iu0.Available structures PDB 2BYG, 2HE2 Identifiers Symbols DLG2; DKFZp781D1854; DKFZp781E0954; FLJ37266; MGC131811; PSD-93; PSD93; chapsyn-110 External IDs OMIM: 603583 MGI: 1344351 HomoloGene: 1046 GeneCards: DLG2 Gene Gene Ontology Molecular function • guanylate kinase activity
• protein bindingCellular component • plasma membrane
• postsynaptic density
• cell junction
• juxtaparanode region of axon
• synapse
• postsynaptic membraneBiological process • synaptic transmission
• sensory perception of painSources: Amigo / QuickGO RNA expression pattern 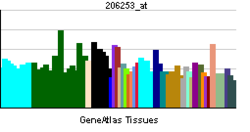
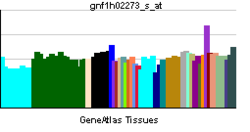
More reference expression data Orthologs Species Human Mouse Entrez 1740 23859 Ensembl ENSG00000150672 ENSMUSG00000052572 UniProt Q15700 O88523 RefSeq (mRNA) NM_001142699.1 NM_011807.2 RefSeq (protein) NP_001136171.1 NP_035937.2 Location (UCSC) Chr 11:
83.17 – 85.34 MbChr 7:
97.65 – 99.6 MbPubMed search [1] [2] Disks large homolog 2 (DLG2) also known as channel-associated protein of synapse-110 (chapsyn-110) or postsynaptic density protein 93 (PSD-93) is a protein that in humans is encoded by the DLG2 gene.[1][2]
Contents
Function
Chapsyn-110/PSD-93 a member of the membrane-associated guanylate kinase (MAGUK) family. The protein forms a heterodimer with a related family member that may interact at postsynaptic sites to form a multimeric scaffold for the clustering of receptors, ion channels, and associated signaling proteins. Alternatively spliced transcript variants encoding distinct isoforms have been described but their full-length nature has yet to be completely determined.[3]
Interactions
DLG2 has been shown to interact with KCNJ12[4] and GRIN2B.[5][6]
References
- ^ Kim E, Cho KO, Rothschild A, Sheng M (December 1996). "Heteromultimerization and NMDA receptor-clustering activity of Chapsyn-110, a member of the PSD-95 family of proteins". Neuron 17 (1): 103–13. doi:10.1016/S0896-6273(00)80284-6. PMID 8755482.
- ^ Stathakis DG, Lee D, Bryant PJ (January 1999). "Fine-scale physical map of the 11q21 region surrounding the human DLG2 locus, the gene encoding Chapsyn-110". Genomics 54 (1): 186–8. doi:10.1006/geno.1998.5527. PMID 9806853.
- ^ "Entrez Gene: DLG2 discs, large homolog 2, chapsyn-110 (Drosophila)". http://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=1740.
- ^ Leonoudakis, Dmitri; Conti Lisa R, Anderson Scott, Radeke Carolyn M, McGuire Leah M M, Adams Marvin E, Froehner Stanley C, Yates John R, Vandenberg Carol A (May 2004). "Protein trafficking and anchoring complexes revealed by proteomic analysis of inward rectifier potassium channel (Kir2.x)-associated proteins". J. Biol. Chem. (United States) 279 (21): 22331–46. doi:10.1074/jbc.M400285200. ISSN 0021-9258. PMID 15024025.
- ^ Inanobe, Atsushi; Fujita Akikazu, Ito Minoru, Tomoike Hitonobu, Inageda Kiyoshi, Kurachi Yoshihisa (June 2002). "Inward rectifier K+ channel Kir2.3 is localized at the postsynaptic membrane of excitatory synapses". Am. J. Physiol., Cell Physiol. (United States) 282 (6): C1396–403. doi:10.1152/ajpcell.00615.2001. ISSN 0363-6143. PMID 11997254.
- ^ Irie, M; Hata Y, Takeuchi M, Ichtchenko K, Toyoda A, Hirao K, Takai Y, Rosahl T W, Südhof T C (September 1997). "Binding of neuroligins to PSD-95". Science (UNITED STATES) 277 (5331): 1511–5. doi:10.1126/science.277.5331.1511. ISSN 0036-8075. PMID 9278515.
Further reading
- Kim E, Niethammer M, Rothschild A, et al. (1995). "Clustering of Shaker-type K+ channels by interaction with a family of membrane-associated guanylate kinases.". Nature 378 (6552): 85–8. doi:10.1038/378085a0. PMID 7477295.
- Brenman JE, Chao DS, Gee SH, et al. (1996). "Interaction of nitric oxide synthase with the postsynaptic density protein PSD-95 and alpha1-syntrophin mediated by PDZ domains.". Cell 84 (5): 757–67. doi:10.1016/S0092-8674(00)81053-3. PMID 8625413.
- Hsueh YP, Kim E, Sheng M (1997). "Disulfide-linked head-to-head multimerization in the mechanism of ion channel clustering by PSD-95.". Neuron 18 (5): 803–14. doi:10.1016/S0896-6273(00)80319-0. PMID 9182804.
- Irie M, Hata Y, Takeuchi M, et al. (1997). "Binding of neuroligins to PSD-95.". Science 277 (5331): 1511–5. doi:10.1126/science.277.5331.1511. PMID 9278515.
- Niethammer M, Valtschanoff JG, Kapoor TM, et al. (1998). "CRIPT, a novel postsynaptic protein that binds to the third PDZ domain of PSD-95/SAP90.". Neuron 20 (4): 693–707. doi:10.1016/S0896-6273(00)81009-0. PMID 9581762.
- Leonard AS, Davare MA, Horne MC, et al. (1998). "SAP97 is associated with the alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptor GluR1 subunit.". J. Biol. Chem. 273 (31): 19518–24. doi:10.1074/jbc.273.31.19518. PMID 9677374.
- Butz S, Okamoto M, Südhof TC (1998). "A tripartite protein complex with the potential to couple synaptic vesicle exocytosis to cell adhesion in brain.". Cell 94 (6): 773–82. doi:10.1016/S0092-8674(00)81736-5. PMID 9753324.
- Brenman JE, Topinka JR, Cooper EC, et al. (1998). "Localization of postsynaptic density-93 to dendritic microtubules and interaction with microtubule-associated protein 1A.". J. Neurosci. 18 (21): 8805–13. PMID 9786987.
- Craven SE, El-Husseini AE, Bredt DS (1999). "Synaptic targeting of the postsynaptic density protein PSD-95 mediated by lipid and protein motifs.". Neuron 22 (3): 497–509. doi:10.1016/S0896-6273(00)80705-9. PMID 10197530.
- Sans N, Petralia RS, Wang YX, et al. (2000). "A developmental change in NMDA receptor-associated proteins at hippocampal synapses.". J. Neurosci. 20 (3): 1260–71. PMID 10648730.
- Garcia RA, Vasudevan K, Buonanno A (2000). "The neuregulin receptor ErbB-4 interacts with PDZ-containing proteins at neuronal synapses.". Proc. Natl. Acad. Sci. U.S.A. 97 (7): 3596–601. doi:10.1073/pnas.070042497. PMC 16285. PMID 10725395. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=16285.
- Husi H, Ward MA, Choudhary JS, et al. (2000). "Proteomic analysis of NMDA receptor-adhesion protein signaling complexes.". Nat. Neurosci. 3 (7): 661–9. doi:10.1038/76615. PMID 10862698.
- Firestein BL, Craven SE, Bredt DS (2001). "Postsynaptic targeting of MAGUKs mediated by distinct N-terminal domains.". Neuroreport 11 (16): 3479–84. doi:10.1097/00001756-200011090-00016. PMID 11095503.
- Haraguchi K, Satoh K, Yanai H, et al. (2001). "The hDLG-associated protein DAP interacts with dynein light chain and neuronal nitric oxide synthase.". Genes Cells 5 (11): 905–911. doi:10.1046/j.1365-2443.2000.00374.x. PMID 11122378.
- DeMarco SJ, Strehler EE (2001). "Plasma membrane Ca2+-atpase isoforms 2b and 4b interact promiscuously and selectively with members of the membrane-associated guanylate kinase family of PDZ (PSD95/Dlg/ZO-1) domain-containing proteins.". J. Biol. Chem. 276 (24): 21594–600. doi:10.1074/jbc.M101448200. PMID 11274188.
- Russwurm M, Wittau N, Koesling D (2002). "Guanylyl cyclase/PSD-95 interaction: targeting of the nitric oxide-sensitive alpha2beta1 guanylyl cyclase to synaptic membranes.". J. Biol. Chem. 276 (48): 44647–52. doi:10.1074/jbc.M105587200. PMID 11572861.
- Weitzdoerfer R, Dierssen M, Fountoulakis M, Lubec G (2002). "Fetal life in Down syndrome starts with normal neuronal density but impaired dendritic spines and synaptosomal structure.". J. Neural Transm. Suppl. (61): 59–70. PMID 11771761.
- Inanobe A, Fujita A, Ito M, et al. (2002). "Inward rectifier K+ channel Kir2.3 is localized at the postsynaptic membrane of excitatory synapses.". Am. J. Physiol., Cell Physiol. 282 (6): C1396–403. doi:10.1152/ajpcell.00615.2001. PMID 11997254.
PDB gallery Categories:- Human proteins
- Chromosome 11 gene stubs
Wikimedia Foundation. 2010.




