- Magnesium fluoride
-
Magnesium fluoride[1] 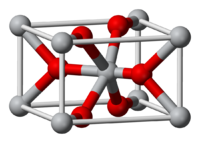 Other namesSellaite
Other namesSellaite
Irtran-1Identifiers CAS number 7783-40-6 
PubChem 24546 ChemSpider 22952 
RTECS number OM3325000 Jmol-3D images Image 1 - [Mg+2].[F-].[F-]
Properties Molecular formula MgF2 Molar mass 62.3018 g/mol Appearance white tetragonal crystals Density 3.148 g/cm3 Melting point 1263 °C [1]
Boiling point 2260 °C (decomp)
Solubility in water 0.013 g/100 mL Solubility product, Ksp 5.16·10-11 Solubility soluble in nitric acid
insoluble in ethanolRefractive index (nD) 1.37397 Structure Crystal structure Rutile (tetragonal), tP6 Space group P42/mnm, No. 136 Thermochemistry Std enthalpy of
formation ΔfHo298-1124.2 kJ·mol-1 Standard molar
entropy So29857.2 J·mol-1·K-1 Specific heat capacity, C 61.6 J·mol-1·K-1 Hazards R-phrases R20, R22 Related compounds Other anions Magnesium chloride
Magnesium bromide
Magnesium iodideOther cations Beryllium fluoride
Calcium fluoride
Strontium fluoride
Barium fluoride fluoride (verify) (what is:
fluoride (verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Magnesium fluoride is an inorganic compound with the formula MgF2. The compound is a white crystalline salt and is transparent over a wide range of wavelengths, with commercial uses in optics.
Contents
Production and structure
Magnesium fluoride is prepared from magnesium oxide with sources of HF such as ammonium bifluoride:
- MgO + (NH4)HF2 → MgF2 + NH3 + H2O
The compound crystallizes as tetragonal birefringent crystals. Its structure is similar to that in rutile, featuring octahedral Mg2+ centers and 3-coordinate fluoride centres.[2]
Magnesium fluoride can also be prepared from the following simple reaction[3]
- MgCl2 + 2((NH4)F) → MgF2 + 2(NH4Cl)
Different with other developments, the use of MgCl2 and (NH4)F allows the fabrication of nano to submicrometer MgF2 with controllable morphology (spherical and cubical)
Uses
Optical properties
Magnesium fluoride is transparent over an extremely wide range of wavelengths. Windows, lenses, and prisms made of this material can be used over the entire range of wavelengths from 0.120 μm (vacuum ultraviolet) to 8.0 μm (infrared). High quality synthetic VUV grade MgF2 is quite expensive, in the region of $3000/kg (2007) but the real cost of optics in this material is due to relatively low volume manufacture. However, with lithium fluoride it is one of the two materials that will transmit in the vacuum ultraviolet range at 121 nm (Lyman alpha) and this is where it finds its application. Lower grade MgF2 is sometimes used in the infrared but here it is inferior to calcium fluoride. MgF2 is tough and works and polishes well, but it is slightly birefringent and should be cut with the optic axis perpendicular to the plane of the window or lens.
The Verdet constant of (MgF2) at 632.8 nm is 0.00810 arcmin G−1 cm−1[4]
Thin layers of MgF2 are frequently applied to the surfaces of optical elements as part of optical coatings such as anti-reflective coatings.
References
- ^ a b Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, FL: CRC Press, pp. 4–67; 1363, ISBN 0-8493-0594-2
- ^ Aigueperse, Jean; Paul Mollard, Didier Devilliers, Marius Chemla, Robert Faron, Renée Romano, Jean Pierre Cuer (2005), "Fluorine Compounds, Inorganic", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a11_307
- ^ A.B.D. Nandiyanto; F. Iskandar; T. Ogi; and K. Okuyama (2010), "Nanometer to Submicrometer Magnesium Fluoride Particles with Controllable Morphology", Langmuir 26 (14): 12260–12266, doi:10.1016/j.actamat.2009.09.004, PMID 20557055.
- ^ J. Chem. Soc., Faraday Trans., 1996, 92, 2753 - 2757. doi:10.1039/FT9969202753
External links
- A java applet showing the effect of MgF2 on a lens
- Infrared windows at Lawrence Berkeley National Laboratory
- National Pollutant Inventory - Fluoride and compounds fact sheet
- Crystran Data Crystran MSDS
Magnesium compounds Categories:- Fluorides
- Magnesium compounds
- Metal halides
- Optical materials
- Inorganic compound stubs
Wikimedia Foundation. 2010.
