- Hydrazine
-
For the antidepressant, see Hydrazine (antidepressant).
Hydrazine 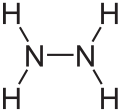
 Systematic nameHydrazine[1]Other names
Systematic nameHydrazine[1]Other namesIdentifiers CAS number 302-01-2 
PubChem 9321 ChemSpider 8960 
UNII 27RFH0GB4R 
EC number 206-114-9 UN number 2029 KEGG C05361 
MeSH Hydrazine ChEBI CHEBI:15571 
ChEMBL CHEMBL1237174 
RTECS number MU7175000 Beilstein Reference 878137 Gmelin Reference 190 3DMet B00770 Jmol-3D images Image 1 - NN
Properties Molecular formula N2H4 Molar mass 32.0452 g mol-1 Exact mass 32.037448138 g mol-1 Appearance Colourless liquid Density 1.021 g cm-3 Melting point 2 °C, 275 K, 35 °F
Boiling point 114 °C, 387 K, 237 °F
log P 0.67 Vapor pressure 1 kP (at 30.7 °C) Acidity (pKa) 8.10[2] Basicity (pKb) 5.90 Refractive index (nD) 1.46044 (at 22 °C) Viscosity 0.876 cP Structure Molecular shape Triangular pyramidal at N Dipole moment 1.85 D[3] Thermochemistry Std enthalpy of
formation ΔfHo29850.63 kJ mol-1 Standard molar
entropy So298121.52. J K-1 mol-1 Hazards MSDS ICSC 0281 GHS pictograms 




GHS signal word DANGER GHS hazard statements H226, H301, H311, H314, H317, H331, H350, H410 GHS precautionary statements P201, P261, P273, P280, P301+310, P305+351+338 EU Index 007-008-00-3 EU classification  T
T  N
NR-phrases R45, R10, R23/24/25, R34, R43, R50/53 S-phrases S53, S45, S60, S61 NFPA 704 Flash point 52 °C Autoignition
temperature24–270 °C Explosive limits 1.8–99.99% LD50 59–60 mg/kg (oral in rats, mice)[4] Related compounds Related compounds Ammonia
 (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Hydrazine is an inorganic compound with the formula N2H4. It is a colourless flammable liquid with an ammonia-like odor. Hydrazine is highly toxic and dangerously unstable unless handled in solution. Approximately 260,000 tons are manufactured annually.[5] Hydrazine is mainly used as a foaming agent in preparing polymer foams, but significant applications also include its uses as a precursor to polymerization catalysts and pharmaceuticals. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in air bags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles to control concentrations of dissolved oxygen in an effort to reduce corrosion.
Contents
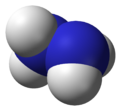
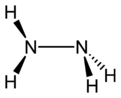
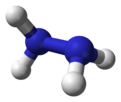
Molecular structure and properties
Hydrazine forms a monohydrate that is more dense (1.032 g/cm3) than the anhydrous material.
Hydrazine can arise via coupling a pair of ammonia molecules by removal of one hydrogen per molecule. Each H2N-N subunit is pyramidal in shape. The N-N distance is 1.45 Å (145 pm), and the molecule adopts a gauche conformation.[6] The rotational barrier is twice that of ethane. These structural properties resemble those of gaseous hydrogen peroxide, which adopts a "skewed" anticlinal conformation, and also experiences a strong rotational barrier.
Hydrazine has basic (alkali) chemical properties comparable to those of ammonia:
- N2H4 + H2O → [N2H5]+ + OH−
with the values:[7]
- Kb = 1.3 x 10−6
- pKa = 8.1
(for ammonia Kb = 1.78 x 10−5)
Hydrazine is difficult to diprotonate:[8]
- [N2H5]+ + H2O → [N2H6]2+ + OH− Kb = 8.4 x 10−16
The heat of combustion of hydrazine in oxygen (air) is 194.1 x 105 J/kg (9345 BTU/lb).[9]
Synthesis and manufacture
Theodor Curtius synthesized free hydrazine for the first time in 1889 via a circuitous route.[10]
Hydrazine is produced in the Olin Raschig process from sodium hypochlorite (the active ingredient in many bleaches) and ammonia, a process announced in 1907. This method relies on the reaction of chloramine with ammonia:[11]
- NH2Cl + NH3 → H2N-NH2 + HCl
Another route of hydrazine synthesis involves oxidation of urea with sodium hypochlorite:[12]
- (H2N)2C=O + NaOCl + 2 NaOH → N2H4 + H2O + NaCl + Na2CO3
Hydrazine can be synthesized from ammonia and hydrogen peroxide in the Pechiney-Ugine-Kuhlmann process, according to the following formula:
- 2NH3 + H2O2 → H2N-NH2 + 2H2O [13]
In the Atofina–PCUK cycle, hydrazine is produced in several steps from acetone, ammonia, and hydrogen peroxide. Acetone and ammonia first react to give the imine followed by oxidation with hydrogen peroxide to the oxaziridine, a three-membered ring containing carbon, oxygen, and nitrogen, followed by ammonolysis to the hydrazone, a process that couples two nitrogen atoms. This hydrazone reacts with one more equivalent of acetone, and the resulting acetone azine is hydrolyzed to give hydrazine, regenerating acetone. Unlike the Raschig process, this process does not produce salt. The PCUK stands for Produits Chimiques Ugine Kuhlmann, a French chemical manufacturer.[14]
Hydrazine can also be produced via the so-called ketazine and peroxide processes.
Hydrazine derivatives
Many substituted hydrazines are known, and several occur naturally. Some examples include
- monomethyl hydrazine, where one of the hydrogen atoms on the hydrazine molecule has been replaced with a methyl group (CH3). By the symmetry of the hydrazine molecule, it does not matter which hydrogen atom is replaced. It is sometimes used as a rocket fuel.
- 1,1-dimethylhydrazine (unsymmetrical dimethylhydrazine, UDMH) and 1,2-dimethylhydrazine (symmetrical dimethylhydrazine) are hydrazines where two hydrogen atoms are replaced by methyl groups. UDMH is easier to manufacture than symmetrical dimethylhydrazine is, and UDMH is a fairly common rocket fuel.
- gyromitrin and agaritine are hydrazine derivatives found in the commercially produced mushroom species Agaricus bisporus. Gyromitrin is metabolized into monomethyl hydrazine.
- Isoniazid, iproniazid, hydralazine, and phenelzine are medications whose molecules contain hydrazine-like structures.
- 2,4-dinitrophenylhydrazine (2,4-DNPH) is commonly used to test for ketones and aldehydes in organic chemistry.
- phenylhydrazine, C6H5NHNH2, the first hydrazine to be discovered.
Applications
The majority use of hydrazine is as a precursor to blowing agents. Specific compounds include azodicarbonamide and azobisisobutyronitrile, which yield 100-200 mL of gas per gram of precursor. In a related application, sodium azide, the gas-forming agent in air bags, is produced from hydrazine by reaction with sodium nitrite.[5]
Hydrazine is also used as a propellant on board space vehicles, and to both reduce the concentration of dissolved oxygen in and control pH of water used in large industrial boilers. The F-16 fighter jet uses hydrazine to fuel the aircraft's emergency power unit.
Precursor to pesticides and pharmaceuticals
Hydrazine is a useful building block in organic synthesis of pharmaceuticals and pesticides. One example is 3-amino-1,2,4-triazole and another is maleic hydrazide. The antitubercular drug isoniazid is prepared from hydrazine.
Hydrazine in biology
Hydrazine is the intermediate in the anaerobic oxidation of ammonia (anammox) process.[15] It is produced by some yeasts and the open ocean bacterium anammox (Brocadia anammoxidans).[16]
Organic chemistry
Hydrazines are part of many organic syntheses, often those of practical significance in pharmaceuticals, such as the antituberculosis medication Isoniazid and the antifungal Fluconazole, as well as in textile dyes and in photography.[5]
Hydrazone formation
Illustrative of the condensation of hydrazine with a simple carbonyl is its reaction with propanone to give the diisopropylidene hydrazine (acetone azine). The latter reacts further with hydrazine to afford the hydrazone:[17]
- 2 (CH3)2CO + N2H4 → 2 H2O + [(CH3)2C=N]2
- [(CH3)2C=N]2 + N2H4 → 2 (CH3)2C=NNH2
The propanone azine is an intermediate in the Atofina-PCUK synthesis. Direct alkylation of hydrazines with alkyl halides in the presence of base affords alkyl-substituted hydrazines, but the reaction is typically inefficient due to poor control on level of substitution (same as in ordinary amines). The reduction of hydrazones to hydrazines present a clean way to produce 1,1-dialkylated hydrazines.
In a related reaction, 2-cyanopyridines react with hydrazine to form amide hydrazides, which can be converted using 1,2-diketones into triazines.
Wolff-Kishner reduction
Hydrazine is used in the Wolff-Kishner reduction, a reaction that transforms the carbonyl group of a ketone or aldehyde into a methylene (or methyl) group via a hydrazone intermediate. The production of the highly stable dinitrogen from the hydrazine derivative helps to drive the reaction.
Heterocyclic chemistry
Being bifunctional, with two amines, hydrazine is a key building block for the preparation of many heterocyclic compounds via condensation with a range of difunctional electrophiles. With 2,4-pentanedione, it condenses to give the 3,5-dimethylpyrazole.[18] In the Einhorn-Brunner reaction hydrazines react with imides to give triazoles.
Sulfonation
Being a good nucleophile, N2H4 can attack sulfonyl halides and acyl halides.[19] The tosylhydrazine also forms hydrazones upon treatment with carbonyls.
Deprotection of phthalimides
Hydrazine is used to cleave N-alkylated phthalimide derivatives. This scission reaction allows phthalimide anion to be used as amine precursor in the Gabriel synthesis.[20]
Reducing agent
Hydrazine is a convenient reductant because the by-products are typically nitrogen gas and water. Thus, it is used as an antioxidant, an oxygen scavenger, and a corrosion inhibitor in water boilers and heating systems. It is also used to reduce metal salts and oxides to the pure metals in electroless nickel plating and plutonium extraction from nuclear reactor waste. Some colour photographic processes also use a weak solution of hydrazine as a stabilizing wash, as it scavenges dye coupler and unreacted silver halides.
Hydrazinium salts
Hydrazine is converted to solid salts by treatment with mineral acids. A common salt is hydrazine sulfate, [N2H5]HSO4, called hydrazinium sulfate.[21] Hydrazine sulfate was investigated as a treatment of cancer-induced cachexia, but proved ineffective.[22]
Hydrazine azide (N5H5), the salt of hydrazine and hydrazoic acid, was of scientific interest, because of its high nitrogen content and explosive properties. Structurally, it is [N2H5]+[N3]−. It decomposes explosively into hydrazine, ammonia and nitrogen gas:[23]
- 12 N5H5 → 3 N2H4 + 16 NH3 + 19 N2
Reaction of N5H5 with sulfuric acid gives quantitative yields of pure hydrazine sulfate and hydrazoic acid.[24]
Other industrial uses
Hydrazine is used in many processes including: production of spandex fibers, as a polymerization catalyst; in fuel cells, solder fluxes; and photographic developers, as a chain extender in urethane polymerizations, and heat stabilizers. In addition, a semiconductor deposition technique using hydrazine has recently been demonstrated, with possible application to the manufacture of thin-film transistors used in liquid crystal displays. Hydrazine in a 70% hydrazine, 30% water solution is used to power the EPU (emergency power unit) on the Lockheed F-16 Fighting Falcon fighter plane. The explosive Astrolite is made by combining hydrazine with ammonium nitrate.
Hydrazine is often used as an oxygen scavenger and corrosion inhibitor in boiler water treatment. However due to the toxicity and certain undesired effects, namely increased rates of flow-accelerated corrosion (FAC)[citation needed], this practice is discouraged.
Rocket fuel
Hydrazine was first used as a rocket fuel during World War II for the Messerschmitt Me 163B (the first rocket-powered fighter plane), under the code name B-Stoff (hydrazine hydrate). When mixed with methanol (M-Stoff) and water it was called C-Stoff.
Hydrazine is also used as a low-power monopropellant for the maneuvering thrusters of spacecraft, and the Space Shuttle's auxiliary power units (APUs). In addition, monopropellant hydrazine-fueled rocket engines are often used in terminal descent of spacecraft. A collection of such engines was used in both Viking program landers as well as the Phoenix lander launched in August 2007.
In all hydrazine monopropellant engines, the hydrazine is passed by a catalyst such as iridium metal supported by high-surface-area alumina (aluminium oxide) or carbon nanofibers,[25] or more recently molybdenum nitride on alumina,[26] which causes it to decompose into ammonia, nitrogen gas, and hydrogen gas according to the following reactions:
- 3 N2H4 → 4 NH3 + N2
- N2H4 → N2 + 2 H2
- 4 NH3 + N2H4 → 3 N2 + 8 H2
These reactions are extremely exothermic (the catalyst chamber can reach 800 °C in a matter of milliseconds,[25]) and they produce large volumes of hot gas from a small volume of liquid hydrazine,[26] making it a fairly efficient thruster propellant with a vacuum specific impulse of about 220 seconds.[27]
Other variants of hydrazine that are used as rocket fuel are monomethylhydrazine, (CH3)NH(NH2) (also known as MMH) and unsymmetrical dimethylhydrazine, (CH3)2N(NH2) (also known as UDMH). These derivatives are used in two-component rocket fuels, often together with nitrogen tetroxide, N2O4, sometimes known as dinitrogen tetroxide. This reaction is extremely exothermic, as a rocket fuel must be, and the burning is also hypergolic, which means that the burning starts without any external ignition source.
Fuel cells
The Italian catalyst manufacturer Acta has proposed using hydrazine as an alternative to hydrogen in fuel cells. The chief benefit of using hydrazine is that it can produce over 200 mW/cm2 more than a similar hydrogen cell without the need to use expensive platinum catalysts. As the fuel is liquid at room temperature, it can be handled and stored more easily than hydrogen. By storing the hydrazine in a tank full of a double-bonded carbon-oxygen carbonyl, the fuel reacts and forms a safe solid called hydrazone. By then flushing the tank with warm water, the liquid hydrazine hydrate is released. Hydrazine has a higher electromotive force of 1.56 V compared to 1.23 V for hydrogen. Hydrazine breaks down in the cell to form nitrogen and hydrogen which bonds with oxygen, releasing water.[28] Hydrazine was used in fuel cells manufactured by Allis-Chalmers Corp., including some that provided electric power in space satellites in the 1960s.
Gun Propellant
A mixture of 63% Hydrazine, 32% Hydrazine Nitrate and 5% water is a standard propellant for experimental bulk-loaded liquid propellant artillery. The propellant mixture above is notable for being one of the most predictable and stable, with a remarkably flat pressure profile during firing. Misfires are usually caused by inadequate ignition. The movement of the shell after a misignition causes a large bubble with a larger ignition surface area, and the greater rate of gas production causes very high pressures, sometimes including catastrophic tube failures (explosions).[29]
Hazards
Hydrazine is highly toxic and dangerously unstable, especially in the anhydrous form. According to the U.S. Environmental Protection Agency:
Symptoms of acute (short-term) exposure to high levels of hydrazine may include irritation of the eyes, nose, and throat, dizziness, headache, nausea, pulmonary edema, seizures, coma in humans. Acute exposure can also damage the liver, kidneys, and central nervous system. The liquid is corrosive and may produce dermatitis from skin contact in humans and animals. Effects to the lungs, liver, spleen, and thyroid have been reported in animals chronically exposed to hydrazine via inhalation. Increased incidences of lung, nasal cavity, and liver tumors have been observed in rodents exposed to hydrazine.[30]
Limit tests for hydrazine in pharmaceuticals suggest that it should be in the low ppm range.[31] Hydrazine may also cause steatosis.[32] At least one human is known to have died, after 6 months of sublethal exposure to hydrazine hydrate.[33]
On February 21, 2008, the United States government destroyed the disabled spy satellite USA 193 with a sea-launched missile, reportedly due to the potential danger of a hydrazine release if it re-entered the Earth's atmosphere intact.[34]
See also
References
- ^ a b "hydrazine - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information. http://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=9321.
- ^ Hall, H.K., J. Am. Chem. Soc., 1957, 79, 5441.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Oxford: Butterworth-Heinemann. ISBN 0080379419.
- ^ Martel, B.; Cassidy, K. (2004). Chemical Risk Analysis: A Practical Handbook. Butterworth–Heinemann. pp. 361. ISBN 1903996651.
- ^ a b c Jean-Pierre Schirmann, Paul Bourdauducq "Hydrazine" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002. doi:10.1002/14356007.a13_177.
- ^ Miessler, Gary L. and Tarr, Donald A. Inorganic Chemistry, Third Edition Pearson Prentice Hall (2004) ISBN 0-13-035471-6.
- ^ Handbook of Chemistry and Physics", 83rd edition, CRC Press, 2002
- ^ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ^ Chemical Hazard Properties Table at NOAA.gov
- ^ Curtius, J. Prakt. Chem. 1889, 39, 107-39.
- ^ Adams, R.; Brown, B. K. (1941), "Hydrazine Sulfate", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv1p0309; Coll. Vol. 1: 309
- ^ "Hydrazine: Chemical product info". chemindustry.ru. http://chemindustry.ru/Hydrazine.php. Retrieved 2007-01-08.
- ^ Chemistry of Petrochemical Processes, 2nd edition, Gulf Publishing Company, 1994-2000, Page 148
- ^ Riegel, Emil Raymond (1992). "Hydrazine". Riegel's Handbook of Industrial Chemistry. p. 192.
- ^ Strous, M., and Jetten, M.S.M. (2004) Anaerobic oxidation of methane and ammonium. Ann Rev Microbiol 58: 99–117.
- ^ Brian Handwerk (9 November 2005). "Bacteria Eat Human Sewage, Produce Rocket Fuel". National Geographic. http://news.nationalgeographic.com/news/2005/11/1109_051109_rocketfuel.html. Retrieved 2007-11-12.
- ^ Day, A. C.; Whiting, M. C., "Acetone Hydrazone", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv6p0010; Coll. Vol. 6: 10
- ^ Wiley, R. H.; Hexner, P. E., "3,5-Dimethylpyrazole", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv4p0351; Coll. Vol. 4: 351
- ^ Friedman, L; Litle, R. L.; Reichle, W. R., "p-Toluenesulfonyl Hydrazide", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv5p1055; Coll. Vol. 5: 1055
- ^ Weinshenker, N. M.; Shen, C. M.; Wong, J. Y. (1988), "Polymeric carbodiimide", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv6p0951; Coll. Vol. 6: 951
- ^ Safety Data Sheet Mallinckrodt
- ^ Gagnon B, Bruera E (May 1998). "A review of the drug treatment of cachexia associated with cancer". Drugs 55 (5): 675–88. doi:10.2165/00003495-199855050-00005. PMID 9585863.
- ^ G. B. Manelis (2003). Thermal decomposition and combustion of explosives and propellants. CRC Press. p. 235. ISBN 0415299845.
- ^ Klapötke, T.; Peter S. White; Inis C. Tornieporth-Oetting (1996). "Reaction of hydrazinium azide with sulfuric acid: the X-ray structure of [N2H6][SO4]". Polyhedron 15 (15): 2579–2582. doi:10.1016/0277-5387(95)00527-7.
- ^ a b Vieira, R.; C. Pham-Huu, N. Keller and M. J. Ledoux (2002). "New carbon nanofiber/graphite felt composite for use as a catalyst support for hydrazine catalytic decomposition". Chemical Communications (9): 954–955. doi:10.1039/b202032g.
- ^ a b Chen, Xiaowei; et al. (April 2002). "Catalytic Decomposition of Hydrazine over Supported Molybdenum Nitride Catalysts in a Monopropellant Thruster". Catalysis Letters 79: 21–25. doi:10.1023/A:1015343922044.
- ^ Monopropellant Hydrazine Thrusters
- ^ Liquid asset - News - The Engineer - [News: engineering news, engineering info, latest technology, manufacturing news, manufacturing info, automotive news, aerospace news, materials news, research & development]
- ^ Knapton, John, Stobie, Irvin, Elmore, Les; ARl-TR-81 A review of the Bulk-Loaded Liquid Propellant Gun Program for Possible Relevance to the Electrothermal Chemical Propulsion Program, Army Research Laboratory, March 1993 At Accessed 2011-7-23
- ^ United States Environmental Protection Agency. Hydrazine Hazard Summary-Created in April 1992; Revised in January 2000[1]. Retrieved on February 21, 2008.
- ^ European Pharmacopeia Scientific Notes. Acceptance criteria for levels of hydrazine in substances for pharmaceutical use and analytical methods for its determination[2]. Retrieved on April 22, 2008.
- ^ PHM 450 Course, Spring 2009, Michigan State University
- ^ International Programme on Chemical Safety, Environmental Health Criteria for Hydrazine, Section 9.2.1, dated 1987. Retrieved on February 21, 2008.
- ^ "IEEE Spectrum Online. U.S. Satellite Shootdown". http://spectrum.ieee.org/aug08/6533. Retrieved 2008-08-08.
External links
- The Late Show with Rob! Tonight’s Special Guest: Hydrazine (PDF) — Robert Matunas
- Hydrazine - chemical product info: properties, production, applications.
- Hydrazine toxicity
Hydrazines 4-PTSC • Acylhydrazine • ADH • Adjudin • Agaritine • Benmoxin • Cadralazine • Carbazide • Carbazone • Carbidopa • Carbohydrazide • Daminozide • Dihydralazine • DNPH • Endralazine • Gyromitrin • HBT • Hydralazine • Hydrazide • Hydrazine • Hydrazone • IDH • Iproclozide • Iproniazid • Isocarboxazid • Isoniazid • Mebanazine • Metfendrazine • MMH • Nialamide • Octamoxin • PEH • Phenelzine • Pheniprazine • Phenoxypropazine • Phenylhydrazine • Pimagedine • Pivalylbenzhydrazine • Procarbazine • Safrazine • SDH • Semicarbazide • Semicarbazone • SDMH • Tetrafluorohydrazine • Thiosemicarbazide • Thiosemicarbazone • UDMHCategories:- Hydrazines
- Bases
- Hazardous air pollutants
- Rocket fuels
- Monopropellants
- Corrosion inhibitors
- Monoamine oxidase inhibitors
- Reducing agents
- Nitrogen hydrides
Wikimedia Foundation. 2010.

