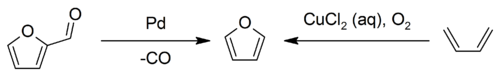- Furan
-
This article is about the chemical industrial compound. For the unintentional contaminant byproduct, see Polychlorinated dibenzofurans. For other uses, see Furan (disambiguation).
Furan 

 FuranOther namesOxole, furfuran, furane (misspelling),[1] divinylene oxide,
FuranOther namesOxole, furfuran, furane (misspelling),[1] divinylene oxide,
1,4-epoxy-1,3-butadiene,
1,4-epoxybuta-1,3-dieneIdentifiers CAS number 110-00-9 
PubChem 8029 ChemSpider 7738 
KEGG C14275 
ChEBI CHEBI:35559 
ChEMBL CHEMBL278980 
Jmol-3D images Image 1 - c1ccoc1
Properties Molecular formula C4H4O Molar mass 68.07 g/mol Appearance colorless, volatile liquid Density 0.936 g/mL Melting point -85.6 °C
Boiling point 31.3 °C
Hazards Flash point -69 °C Related compounds Related heterocycles Pyrrole
ThiopheneRelated compounds Tetrahydrofuran (THF)
2,5-Dimethylfuran
Benzofuran
Dibenzofuran (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen. The class of compounds containing such rings are also referred to as furans.
Furan is a colorless, flammable, highly volatile liquid with a boiling point close to room temperature. It is toxic and may be carcinogenic. Furan is used as a starting point to other specialty chemicals.[2]
Contents
History
The name furan comes from the Latin furfur, which means bran.[3] The first furan derivative to be described was 2-furoic acid, by Carl Wilhelm Scheele in 1780. Another important derivative, furfural, was reported by Johann Wolfgang Döbereiner in 1831 and characterised nine years later by John Stenhouse. Furan itself was first prepared by Heinrich Limpricht in 1870, although he called it tetraphenol.[4][5]
Production
Industrially, furan is manufactured by the palladium-catalyzed decarbonylation of furfural, or by the copper-catalyzed oxidation of 1,3-butadiene:[2]
In the laboratory, furan can be obtained from furfural by oxidation to furan-2-carboxylic acid, followed by decarboxylation.[6]. It can also be prepared directly by thermal decomposition of pentose-containing materials, cellulosic solids especially pine-wood.
Synthesis of furans
The Feist-Benary synthesis is a classic way to synthesize furans, although many syntheses have been developed.[7] One of the simplest synthesis methods for furans is the reaction of 1,4-diketones with phosphorus pentoxide (P2O5) in the Paal-Knorr Synthesis. The thiophene formation reaction of 1,4-diketones with Lawesson's reagent also forms furans as side products. 2,4-Disubstituted furans can be synthesized by sulfone-mediated cyclization of 1,3-diketones[8]
Chemistry
Furan is aromatic because one of the lone pairs of electrons on the oxygen atom is delocalized into the ring, creating a 4n+2 aromatic system (see Hückel's rule) similar to benzene. Because of the aromaticity, the molecule is flat and lacks discrete double bonds. The other lone pair of electrons of the oxygen atom extends in the plane of the flat ring system. The sp2 hybridization is to allow one of the lone pairs of oxygen to reside in a p orbital and thus allow it to interact within the pi-system.
Due to its aromaticity, furan's behavior is quite dissimilar to that of the more typical heterocyclic ethers such as tetrahydrofuran.
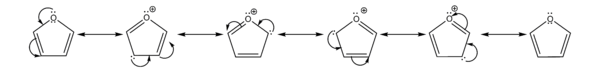
- It is considerably more reactive than benzene in electrophilic substitution reactions, due to the electron-donating effects of the oxygen heteroatom. Examination of the resonance contributors shows the increased electron density of the ring, leading to increased rates of electrophilic substitution.[9]
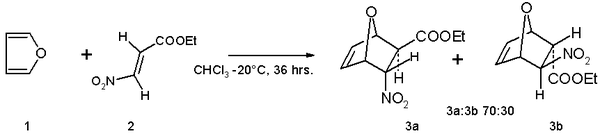
- Furan serves as a diene in Diels-Alder reactions with electron-deficient dienophiles such as ethyl (E)-3-nitroacrylate.[10] The reaction product is a mixture of isomers with preference for the endo isomer:
- Hydrogenation of furans affords sequentially dihydrofurans and tetrahydrofurans.
- In the Achmatowicz reaction, furans converted to dihydropyran compounds.
See also
- Simple aromatic rings
- Furanose
- bs4994 Furan resin as thermoset FRP for chemical process plant equipments
- Furantetracarboxylic acid
- Furanoflavonoid
References
- ^ Webster's Online Dictionary
- ^ a b H. E. Hoydonckx, W. M. Van Rhijn, W. Van Rhijn, D. E. De Vos, P. A. Jacobs (2005), "Furfural and Derivatives", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a12_119.pub2
- ^ Alexander Senning. Elsevier's Dictionary of Chemoetymology. Elsevier, 2006. ISBN 0444522395.
- ^ Limpricht, H. (1870). "Ueber das Tetraphenol C4H4O". Berichte der deutschen chemischen Gesellschaft 3 (1): pp. 90–91. doi:10.1002/cber.18700030129.
- ^ Rodd, Ernest Harry (1971). Chemistry of Carbon Compounds: A Modern Comprehensive Treatise. Elsevier.
- ^ Wilson, W.C. (1941), "Furan", Org. Synth., http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv1p0274; Coll. Vol. 1: 274
- ^ Hou XL, Cheung HY, Hon TY, Kwan PL, Lo TH, Tong SY, Wong HNC (1998). ""Regioselective syntheses of substituted furans"". Tetrahedron 54 (10): 1955–2020. doi:10.1016/S0040-4020(97)10303-9.
- ^ Haines, N. R.; VanZanten, A. N.; Cuneo, A. A.; Miller, J. R.; Andrews, W. J.; Carlson, D. A.; Harrington, R. M.; Kiefer, A. M.; Mason, J. D.; Pigza, J. A.; Murphree, S. S. [1] Journal of Organic Chemistry, Volume 76, p. 8131 (2011).
- ^ Bruice, Paula Y. (2007). Organic Chemistry (Fifth ed.). Upper Saddle River, NJ: Pearson Prentice Hall. ISBN 0-13-196316-3.
- ^ Masesane I, Batsanov A, Howard J, Modal R, Steel P (2006). "The oxanorbornene approach to 3-hydroxy, 3,4-dihydroxy and 3,4,5-trihydroxy derivatives of 2-aminocyclohexanecarboxylic acid". Beilstein Journal of Organic Chemistry 2 (9): 9. doi:10.1186/1860-5397-2-9. PMC 1524792. PMID 16674802. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=1524792.
External links
Categories:- Furans
- IARC Group 2B carcinogens
Wikimedia Foundation. 2010.