- Density of air
-
The density of air, ρ (Greek: rho) (air density), is the mass per unit volume of Earth's atmosphere, and is a useful value in aeronautics and other sciences. Air density decreases with increasing altitude, as does air pressure. It also changes with variances in temperature or humidity. At sea level and at 15°C according to ISA (International Standard Atmosphere), air has a density of approximately 1.275 kg/m3.
Contents
Relationships
Temperature and pressure
The density of dry air can be calculated using the ideal gas law, expressed as a function of temperature and pressure:
where ρ is the air density, p is absolute pressure, Rspecific is the specific gas constant for dry air, and T is absolute temperature.
The specific gas constant for dry air is 287.058 J/(kg·K) in SI units, and 53.35 (ft·lbf)/(lbm·R) in United States customary and Imperial units.
Therefore:
- At IUPAC standard temperature and pressure (0 °C and 100 kPa), dry air has a density of 1.2754 kg/m3.
- At 20 °C and 101.325 kPa, dry air has a density of 1.2041 kg/m3.
- At 70 °F and 14.696 psi, dry air has a density of 0.074887 lbm/ft3.
The following table illustrates the air density - temperature relationship at 1 atm or 101.325 kPa:
Effect of temperature Temperature Speed of sound Density of air Acoustic impedance  in °C
in °Cc in m·s−1 ρ in kg·m−3 Z in N·s·m−3 +35 351.96 1.1455 403.2 +30 349.08 1.1644 406.5 +25 346.18 1.1839 409.4 +20 343.26 1.2041 413.3 +15 340.31 1.2250 416.9 +10 337.33 1.2466 420.5 +5 334.33 1.2690 424.3 ±0 331.30 1.2920 428.0 -5 328.24 1.3163 432.1 -10 325.16 1.3413 436.1 -15 322.04 1.3673 440.3 -20 318.89 1.3943 444.6 -25 315.72 1.4224 449.1 Water vapor
The addition of water vapor to air (making the air humid) reduces the density of the air, which may at first appear contrary to logic.
This occurs because the molecular mass of water (18 g/mol) is less than the molecular mass of dry air (around 29 g/mol). For any gas, at a given temperature and pressure, the number of molecules present is constant for a particular volume (see Avogadro's Law). So when water molecules (vapor) are added to a given volume of air, the dry air molecules must decrease by the same number, to keep the pressure or temperature from increasing. Hence the mass per unit volume of the gas (its density) decreases.
The density of humid air may be calculated as a mixture of ideal gases. In this case, the partial pressure of water vapor is known as the vapor pressure. Using this method, error in the density calculation is less than 0.2% in the range of −10 °C to 50 °C. The density of humid air is found by:
where:
 Density of the humid air (kg/m³)
Density of the humid air (kg/m³)- pd = Partial pressure of dry air (Pa)
- Rd = Specific gas constant for dry air, 287.058 J/(kg·K)
- T = Temperature (K)
- pv = Pressure of water vapor (Pa)
- Rv = Specific gas constant for water vapor, 461.495 J/(kg·K)
The vapor pressure of water may be calculated from the saturation vapor pressure and relative humidity. It is found by:
Where:
- pv = Vapor pressure of water
- ϕ = Relative humidity
- psat = Saturation vapor pressure
The saturation vapor pressure of water at any given temperature is the vapor pressure when relative humidity is 100%. A simplification of the regression [1] used to find this, can be formulated as:
Note:
- This will give a result in mbar (millibar), 1 mbar = 0.001 bar = 0.1 kPa = 100 Pa
- pd is found considering partial pressure, resulting in:
Where p simply notes the absolute pressure in the observed system.
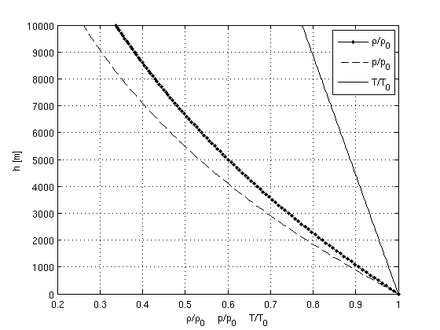
Altitude
To calculate the density of air as a function of altitude, one requires additional parameters. They are listed below, along with their values according to the International Standard Atmosphere, using the universal gas constant instead of the specific one:
- sea level standard atmospheric pressure p0 = 101.325 kPa
- sea level standard temperature T0 = 288.15 K
- Earth-surface gravitational acceleration g = 9.80665 m/s2.
- temperature lapse rate L = 0.0065 K/m
- universal gas constant R = 8.31447 J/(mol·K)
- molar mass of dry air M = 0.0289644 kg/mol
Temperature at altitude h meters above sea level is given by the following formula (only valid inside the troposphere):
The pressure at altitude h is given by:
Density can then be calculated according to a molar form of the original formula:
where M is molar mass, R is the ideal gas constant, and T is absolute temperature.
See also
- International Standard Atmosphere
- U.S. Standard Atmosphere
- NRLMSISE-00
- Air density, density altitude, grains of water calculator by region
References
External links
Categories:
Wikimedia Foundation. 2010.