- Ephedrine
-
Ephedrine 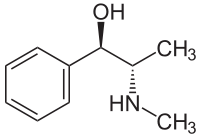
Systematic (IUPAC) name (R*,S*)-2-(methylamino)-1-phenylpropan-1-ol Clinical data AHFS/Drugs.com monograph Pregnancy cat. A(AU) A(US) Legal status Prescription Only (S4) (AU) Schedule VI (CA) P (UK) OTC (US) Routes oral, IV, IM, SC Pharmacokinetic data Bioavailability 85% Metabolism minimal hepatic Half-life 3–6 hours Excretion 22-99% renal Identifiers CAS number 299-42-3 
ATC code C01CA26 R01AA03 R01AB05 R03CA02 S01FB02 QG04BX90 PubChem CID 5032 DrugBank DB01364 ChemSpider 8935 
UNII GN83C131XS 
KEGG D00124 
ChEBI CHEBI:15407 
ChEMBL CHEMBL211456 
Chemical data Formula C10H15NO Mol. mass 165.23 SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Ephedrine (
 /ɨˈfɛdrɪn/ or /ˈɛfɨdriːn/; not to be confused with ephedrone) is a sympathomimetic amine commonly used as a stimulant, appetite suppressant, concentration aid, decongestant, and to treat hypotension associated with anaesthesia.
/ɨˈfɛdrɪn/ or /ˈɛfɨdriːn/; not to be confused with ephedrone) is a sympathomimetic amine commonly used as a stimulant, appetite suppressant, concentration aid, decongestant, and to treat hypotension associated with anaesthesia.Ephedrine is similar in structure to the (semi-synthetic) derivatives amphetamine and methamphetamine. Chemically, it is an alkaloid derived from various plants in the genus Ephedra (family Ephedraceae). It works mainly by increasing the activity of noradrenaline on adrenergic receptors.[1] It is most usually marketed in the hydrochloride and sulfate forms.
In traditional Chinese medicines, the herb má huáng (麻黄, Ephedra sinica) contains ephedrine and pseudoephedrine as its principal active constituents. The same is true of other herbal products containing extracts from Ephedra species.
Contents
History
Nagayoshi Nagai, a Japanese chemist, was the first person to isolate ephedrine from Ephedra distachya (syn. Ephedra vulgaris) in 1885.
Chemistry
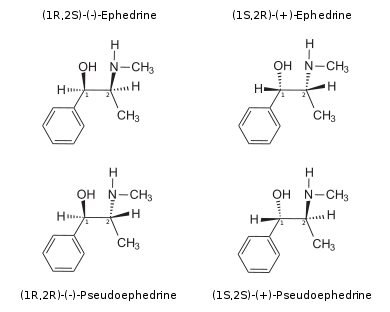
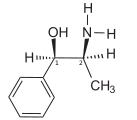
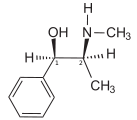
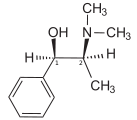
Ephedrine exhibits optical isomerism and has two chiral centres, giving rise to four stereoisomers. By convention the enantiomers with opposite stereochemistry around the chiral centres (1R,2S and 1S,2R) are designated ephedrine, while pseudoephedrine has same stereochemistry around the chiral carbons (1R,2R and 1S,2S).
Ephedrine is a substituted amphetamine and a structural methamphetamine analogue. It differs from methamphetamine only by the presence of a hydroxyl (OH). Amphetamines, however, are more potent and have additional biological effects.
The isomer which is marketed is (–)-(1R,2S)-ephedrine.[2]
Ephedrine hydrochloride has a melting point of 187-188°C.[3]
Nomenclature
The dextrorotary (+)- or d- enantiomer is (1S,2R)-Ephedrine, whereas the levorotating (−)- or l- form is (1R,2S)-Ephedrine.
In the outdated d/l system (+)-Ephedrine is also referred to as l-Ephedrine and (—)-Ephedrine as d-Ephedrine (in the Fisher projection then the phenylring is drawn at bottom).[2][4]
Often the d/l system (with small caps) and the d/l system (with lower-case) are confused. The result is that the levorotary l-Ephedrine is wrongly named l-Ephedrine and the dextrorotary d-Pseudoephedrine (the diastereomer) wrongly d-Pseudoephedrine.
The IUPAC names of the two enantiomers are (1R,2S)- respectively (1S,2R)-2-methylamino-1-phenylpropan-1-ol. A synonym is erythro-Ephedrine.
Agricultural sources
Raw materials for the production of ephedrine and traditional Chinese medicine are produced in China on a large scale. In 2007, companies produced for export extract US$13 million worth of ephedrine from 30,000 tons of ephedra annually, 10 times the amount that is used in traditional Chinese medicine.[5]
Synthetic sources
Most of the L-ephedrine produced today for official medical use is made synthetically as the extraction and isolation process from Herba Ephedra is tedious and no longer cost effective.[6]
Mechanism of action
Ephedrine is a sympathomimetic amine. The principal mechanism of its action relies on its indirect stimulation of the adrenergic receptor system, which is part of the sympathetic nervous system (SNS), by increasing the activity of noradrenaline at the post-synaptic α- and β-receptors.[1] The presence of direct interactions with α-receptors is unlikely, but still controversial.[7][8][9] L-Ephedrine, and particularly its stereoisomer norpseudoephedrine (which is also present in Catha edulis) has indirect sympathomimetic effects and due to its ability to cross the blood brain barrier, it is a CNS stimulant similar to amphetamines but less pronounced, as it releases noradrenaline and dopamine in the substantia nigra.[10]
The presence of an N-methyl group decreases binding affinities at α-receptors, compared with norephedrine. On the other hand ephedrine binds better than steric orientation of the hydroxyl group is important for receptor binding and functional activity.[7]
- Compounds with decreasing α-receptor affinity
Medical use
Indications
In traditional Chinese medicine, ephedrine has been used in the treatment of asthma and bronchitis for centuries.[11]
Both ephedrine and pseudoephedrine act as a bronchodilator, but pseudoephedrine has considerably less effect. Both also increase blood pressure, with again pseudoephedrine being considerably less effective. [9]
Ephedrine promotes weight loss, specifically fat loss in humans and mice. In mice it is known to stimulate thermogenesis in the brown adipose tissue, however because adult humans have only small amounts of brown fat it is assumed that thermogenesis takes place mostly in the skeletal muscle. Ephedrine also decreases gastric emptying. Methylxanthines like caffeine and theophylline have a synergistic effect with ephedrine with respect to weight loss, and so does aspirin. This led to creation and marketing of compound products.[12] One of them is known as the ECA stack, containing caffeine and aspirin besides ephedrine and is a popular supplement taken by body builders to cut down body fat before a competition.[citation needed]
For many years, the US Coast Guard recommended ephedrine together with an equal 25 mg dose of promethazine to its sailors to combat seasickness. Promethazine manages nausea and ephedrine fights the ensuing drowsiness. Commonly referred to as the Coast Guard cocktail, ephedrine may still be available for prescription for this purpose.[citation needed]
Adverse effects
Adverse drug reactions (ADRs) are more common with systemic administration (e.g. injection or oral administration) compared to topical administration (e.g. nasal instillations). ADRs associated with ephedrine therapy include:[13]
- Cardiovascular: tachycardia, cardiac arrhythmias, angina pectoris, vasoconstriction with hypertension
- Dermatological: flushing, sweating, acne vulgaris
- Gastrointestinal: anorexia, nausea
- Genitourinary: decreased urination due to vasoconstriction of renal arteries. Also, difficulty urinating is not uncommon, as alpha-agonists such as ephedrine constrict the internal urethral sphincter, mimicking the effects of sympathetic nervous system stimulation.
- Nervous system: restlessness, confusion, insomnia, mild euphoria, mania/hallucinations (rare except in previously existing psychiatric conditions), delusions, formication (may be possible, but lacks documented evidence) paranoia, hostility, panic, agitation
- Respiratory: dyspnea, pulmonary edema
- Miscellaneous: dizziness, headache, tremor, hyperglycemic reactions, dry mouth
The neurotoxicity of l-Ephedrine is disputed. [14]
Contraindications
Ephedrine should not be used in conjunction with certain antidepressants, namely SNRIs (serotonin-norepinephrine re-uptake inhibitors), as this increases the risk of the above symptoms due to excessive serum levels of norepinephrine.
Bupropion is an example of an antidepressant with an amphetamine-like structure similar to ephedrine, and it is known as an NDRI (norepinephrine-dopamine re-uptake inhibitor). It has an action which bears more resemblance to amphetamine than to fluoxetine in that its primary mode of therapeutic action involves norepinephrine and to a lesser degree dopamine, but it also releases some serotonin from presynaptic clefts. It should not be used with ephedrine as it may increase the likelihood of the above side effects.
Ephedrine should be used with caution in patients with inadequate fluid replacement, impaired adrenal function, hypoxia, hypercapnia, acidosis, hypertension, hyperthyroidism, prostatic hypertrophy, diabetes mellitus, cardiovascular disease, during delivery if maternal BP > 130/80 mmHg, and lactation.[15]
Contraindications for the use of ephedrine include: closed angle glaucoma, phaeochromocytoma, asymmetric septal hypertrophy (idiopathic hypertrophic subaortic stenosis), concomitant or recent (previous 14 days) monoamine oxidase inhibitor (MAOI) therapy, general anaesthesia with halogenated hydrocarbons (particularly halothane), tachyarrhythmias or ventricular fibrillation, hypersensitivity to ephedrine or other stimulants.
Ephedrine should not be used at any time during pregnancy unless specifically indicated by a qualified physician and only when other options are unavailable.[15]
Recreational and illicit use
Anecdotal reports have suggested that ephedrine helps studying, thinking, or concentrating to a greater extent than caffeine. Some students and some white-collar workers have used ephedrine (or Ephedra-containing herbal supplements) for this purpose, as well as some professional athletes and weightlifters. It is common for many athletes to use stimulants while exercising. Such use of ephedrine has been associated with stimulant dependence, as well as deaths from heatstroke in athletes and circulatory problems such as aortic aneurysm in weightlifters, though these side effects are rare.
As a phenylethylamine, ephedrine has a similar chemical structure to amphetamines and is a methamphetamine analogue having the methamphetamine structure with a hydroxyl group at the β position. Because of ephedrine's structural similarity to methamphetamine it can be used to create methamphetamine using chemical reduction in which ephedrine's hydroxy group is removed; this has made ephedrine a highly sought-after chemical precursor in the illicit manufacture of methamphetamine. The most popular method for reducing ephedrine to methamphetamine is similar to the Birch reduction, in that it uses anhydrous ammonia and lithium metal in the reaction. The second most popular method uses red phosphorus, iodine, and ephedrine in the reaction.
In E for Ecstasy[16] (a book examining the uses of the drug MDMA in the UK) the writer, activist and Ecstasy advocate Nicholas Saunders highlighted test results showing that certain consignments of the drug also contained ephedrine. Consignments of Ecstasy known as "Strawberry" contained what Saunders described as a "potentially dangerous combination of ketamine, ephedrine and selegiline," as did a consignment of "Sitting Duck" Ecstasy tablets.[17]
Through oxidation, ephedrine can be easily synthesized into methcathinone. Ephedrine is listed as a Table I precursor under the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances.[18]
Detection of use
Ephedrine may be quantitated in blood, plasma or urine to monitor possible abuse by athletes, confirm a diagnosis of poisoning or assist in a medicolegal death investigation. Many commercial immunoassay screening tests directed at the amphetamines cross-react appreciably with ephedrine, but chromatographic techniques can easily distinguish ephedrine from other phenethylamine derivatives. Blood or plasma ephedrine concentrations are typically in the 20-200 µg/L range in persons taking the drug therapeutically, 300-3000 µg/L in abusers or poisoned patients and 3–20 mg/L in cases of acute fatal overdosage. The current WADA limit for ephedrine in an athlete's urine is 10 µg/L.[19][20][21]
Other uses
In chemical synthesis, ephedrine is used in bulk quantities to produce chiral auxiliary groups.
Legality
Canada
Ephedrine is sold as an oral nasal decongestant in 8 mg pills, OTC.
USA
Ephedrine distribution is controlled by the government, and pharmacists may refuse to sell it to customers without a prescription.
In 1997, the FDA proposed a regulation on ephedra (the herb from which ephedrine is obtained), which limited an ephedra dose to 8 mg (of active ephedrine) with no more than 24 mg per day.[22] This proposed rule was withdrawn in part in 2000 because of "concerns regarding the agency's basis for proposing a certain dietary ingredient level and a duration of use limit for these products."[23] In 2004, the FDA created a ban on ephedrine alkaloids that are marketed for reasons other than asthma, colds, allergies, other disease, or traditional Asian use.[24] On April 14, 2005, the U.S. District Court for the District of Utah ruled that the FDA did not have proper evidence that low dosages of ephedrine alkaloids are actually unsafe,[2] but on August 17, 2006, the U.S. Court of Appeals for the Tenth Circuit in Denver upheld the FDA's final rule declaring all dietary supplements containing ephedrine alkaloids adulterated, and therefore illegal for marketing in the United States. [3] Furthermore, ephedrine is banned by NCAA, MLB, NFL, and PGA TOUR [25] Ephedrine is, however, still legal in many applications outside of dietary supplements. However, purchasing is currently limited and monitored, with specifics varying from state to state.
The House passed the Combat Methamphetamine Epidemic Act of 2005 as an amendment to the renewal of the USA PATRIOT Act. Signed into law by president George W. Bush on March 6, 2006, the act amended the US Code (21 USC 830) concerning the sale of ephedrine-containing products. The federal statute included the following requirements for merchants who sell these products:
- A retrievable record of all purchases identifying the name and address of each party to be kept for two years
- Required verification of proof of identity of all purchasers
- Required protection and disclosure methods in the collection of personal information
- Reports to the Attorney General of any suspicious payments or disappearances of the regulated products
- Non-liquid dose form of regulated product may only be sold in unit dose blister packs
- Regulated products are to be sold behind the counter or in a locked cabinet in such a way as to restrict access
- Daily sales of regulated products not to exceed 3.6 grams without regard to the number of transactions
- Monthly sales not to exceed 9 grams of pseudoephedrine base in regulated products
The law gives similar regulations to mail-order purchases, except the monthly sales limit is only 7.5 grams.
As a pure herb or tea, má huáng, containing ephedrine, is still sold legally in the USA. The law restricts/prohibits its being sold as a dietary supplement(pill) or as an ingredient/additive to other products, like diet pills.
UK
In the UK ephedrine is regulated as a P medicine: it may only be lawfully supplied within a registered pharmacy and while a pharmacist is present. It is not a currently Controlled Drug under the Misuse of Drugs Act.
South Africa
In South Africa ephedrine was rescheduled to Schedule 6 on 27 May 2008,[26] which makes the substance legal to possess but available via a prescription only.
Synthesis
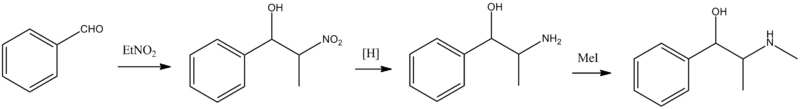
Ephedrine, L-erythro-1-phenyl-2-methylaminopropanol-1, is synthesized from benzaldehyde in a few different ways. According to the first, benzaldehyde is condensed with nitroethane, giving 2-methyl-2-nitro-1-phenylethanol, which is reduced to 2-methyl-2-amino-1-phenylethanol. The necessary L-isomer is isolated from the mixture of isomers by crystallization. Methylation of this gives ephedrine.
- Spath, K. Gohring, Monatsh. Chem., 41, 319 (1920).
- Manske, R. H. F.; Johnson, T. B. (1929). Journal of the American Chemical Society 51 (2): 580. doi:10.1021/ja01377a032.
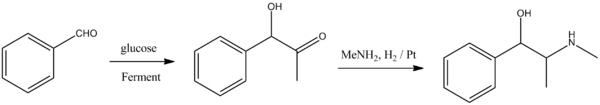
The second method consists of the fermentation of glucose by yeast carboligase in the presence of benzaldehyde, which during the process turns into (–)-1-phenyl-2-ketopropanol. This is reduced by hydrogen in the presence of methylamine, to give the desired ephedrine.
- W. Klawehn, G. Hilderbrandt, U.S. Patent 1,956,950 (1934).
- Merck Chem. Fab. E, DE 469782 (1926).
See also
References
- ^ a b Merck Manuals > EPHEDrine Last full review/revision January 2010
- ^ a b Martindale (1989). Edited by Reynolds JEF. ed. Martindale: The complete drug reference (29th ed.). London: Pharmaceutical Press. ISBN 0-85369-210-6.
- ^ Budavari S, editor. The Merck Index: An encyclopedia of chemicals, drugs, and biologicals, 12th edition. Whitehouse Station: Merck
- ^ Popat N. Patil, A. Tye and J.B. LaPidus A pharmacological study of the ephedrine isomers JPET May 1965 vol. 148, no. 2, pp. 158-168. Full PDF
- ^ Long, Professor. http://www.chinadialogue.net/article/show/single/en/692-Chinese-medicine-s-great-waste-of-resources
- ^ Chemically Synthesized Ephedrine Put Into Mass Production in China
- ^ a b Guoyi Ma, et al. Pharmacological Effects of Ephedrine Alkaloids on Human {alpha}1- and {alpha}2-Adrenergic Receptor Subtypes J. Pharmacol. Exp. Ther.; nr. 322 pp. 214-221 (july 2007) PDF
- ^ Shigeaki Kobayashi, et al. The Sympathomimetic Actions of l-Ephedrine and d-Pseudoephedrine: Direct Receptor Activation or Norepinephrine Release? Anesth Analg 2003; 97, pp.1239-1245.
- ^ a b Drew, et al. Comparison of the effects of D-(-)-ephedrine and L-(+)-pseudoephedrine on the cardiovascular and respiratory systems in man. Br J Clin Pharmacol. 1978; 6, pp 221-225. PDF
- ^ Munhall AC, Johnson SW. (2006). “Dopamine-mediated actions of ephedrine in the rat substantia nigra.” Brain Res. 1069 (1 ): 96-103. PMID: 16386715
- ^ Ford MD, Delaney KA, Ling LJ, Erickson T, editors. Clinical Toxicology. Philadelphia: WB Saunders; 2001. ISBN 0-7216-5485-1 Research Laboratories; 1996. ISBN 0-911910-12-3
- ^ George A. Bray; Claude Bouchard (2004). Handbook of obesity. CRC Press. pp. 494–496. ISBN 9780824747732. http://books.google.com/books?id=YVQPOfKYJhUC&pg=PA494.
- ^ Joint Formulary Committee. British National Formulary, 47th edition. London: British Medical Association and Royal Pharmaceutical Society of Great Britain; 2004. ISBN 0853695873
- ^ Txsci.oxfordjournals (2000).
- ^ a b Mayne Pharma. Ephedrine sulfate injection DBL (Approved Product Information). Melbourne: Mayne Pharma; 2004
- ^ Saunders, N., & Heron, L., (1993) E for Ecstasy (Paperback), N. Saunders, London. (ISBN 0950162884)
- ^ See: [1] for details online.
- ^ Microsoft Word - RedListE2007.doc
- ^ Schier JG, Traub SJ, Hoffman RS, Nelson LS. Ephedrine-induced cardiac ischemia: exposure confirmed with a serum level. Clin. Toxicol. 41: 849-853, 2003.
- ^ WADA. The World Anti-Doping Code, World Anti-Doping Agency, Montreal, Canada, 2010. url
- ^ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 542-544.
- ^ Federal Register: June 4, 1997 (Volume 62, Number 107): Dietary Supplements Containing Ephedrine Alkaloids; Proposed Rule
- ^ Federal Register: April 3, 2000 (Volume 65, Number 64): Dietary Supplements Containing Ephedrine Alkaloids; Withdrawal in Part
- ^ Federal Register: February 11, 2004 (Volume 69, Number 28): Final Rule Declaring Dietary Supplements Containing Ephedrine Alkaloids Adulterated Because They Present an Unreasonable Risk; Final Rule
- ^ http://www.drugfreesport.com/drug-resources/faq.asp
- ^ http://www.doh.gov.za/docs/pr/2008/pr0527.html
Decongestants and other nasal preparations (R01) Topical Sympathomimetics, plainCyclopentamine • Ephedrine • Phenylephrine • Oxymetazoline • Tetryzoline • Xylometazoline • Naphazoline • Tramazoline • Metizoline • Tuaminoheptane • Fenoxazoline • Tymazoline • EpinephrineAntiallergic agents,
excluding corticosteroidsSpaglumic acid
histamine antagonists (Levocabastine, Antazoline, Thonzylamine)
mast cell stabilizer (some are also antihistamines) (Cromoglicic acid, Nedocromil, Azelastine, Olopatadine, Lodoxamide)Other nasal preparationsCafaminol • Calcium hexamine thiocyanate • Retinol • Ipratropium bromide • Ritiometan • Mupirocin • Hexamidine • Framycetin • Hyaluronic acid • Eucalyptus oilSystemic use:
SympathomimeticsOphthalmologicals: mydriatics and cycloplegics (S01F) Anticholinergics/antimuscarinics Sympathomimetics Phenylephrine - Ephedrine - IbopamineM: EYE
anat(g/a/p)/phys/devp/prot
noco/cong/tumr, epon
proc, drug(S1A/1E/1F/1L)
Adrenergics Receptor ligands Agonists: 5-FNE • 6-FNE • Amidephrine • Anisodamine • Anisodine • Cirazoline • Dipivefrine • Dopamine • Ephedrine • Epinephrine (Adrenaline) • Etilefrine • Ethylnorepinephrine • Indanidine • Levonordefrin • Metaraminol • Methoxamine • Methyldopa • Midodrine • Naphazoline • Norepinephrine (Noradrenaline) • Octopamine • Oxymetazoline • Phenylephrine • Phenylpropanolamine • Pseudoephedrine • Synephrine • Tetrahydrozoline
Antagonists: Abanoquil • Adimolol • Ajmalicine • Alfuzosin • Amosulalol • Arotinolol • Atiprosin • Benoxathian • Buflomedil • Bunazosin • Carvedilol • CI-926 • Corynanthine • Dapiprazole • DL-017 • Domesticine • Doxazosin • Eugenodilol • Fenspiride • GYKI-12,743 • GYKI-16,084 • Indoramin • Ketanserin • L-765,314 • Labetalol • Mephendioxan • Metazosin • Monatepil • Moxisylyte (Thymoxamine) • Naftopidil • Nantenine • Neldazosin • Nicergoline • Niguldipine • Pelanserin • Phendioxan • Phenoxybenzamine • Phentolamine • Piperoxan • Prazosin • Quinazosin • Ritanserin • RS-97,078 • SGB-1,534 • Silodosin • SL-89.0591 • Spiperone • Talipexole • Tamsulosin • Terazosin • Tibalosin • Tiodazosin • Tipentosin • Tolazoline • Trimazosin • Upidosin • Urapidil • Zolertine
* Note that many TCAs, TeCAs, antipsychotics, ergolines, and some piperazines like buspirone, trazodone, nefazodone, etoperidone, and mepiprazole all antagonize α1-adrenergic receptors as well, which contributes to their side effects such as orthostatic hypotension.Agonists: (R)-3-Nitrobiphenyline • 4-NEMD • 6-FNE • Amitraz • Apraclonidine • Brimonidine • Cannabivarin • Clonidine • Detomidine • Dexmedetomidine • Dihydroergotamine • Dipivefrine • Dopamine • Ephedrine • Ergotamine • Epinephrine (Adrenaline) • Esproquin • Etilefrine • Ethylnorepinephrine • Guanabenz • Guanfacine • Guanoxabenz • Levonordefrin • Lofexidine • Medetomidine • Methyldopa • Mivazerol • Naphazoline • Norepinephrine (Noradrenaline) • Phenylpropanolamine • Piperoxan • Pseudoephedrine • Rilmenidine • Romifidine • Talipexole • Tetrahydrozoline • Tizanidine • Tolonidine • Urapidil • Xylazine • Xylometazoline
Antagonists: 1-PP • Adimolol • Aptazapine • Atipamezole • BRL-44408 • Buflomedil • Cirazoline • Efaroxan • Esmirtazapine • Fenmetozole • Fluparoxan • GYKI-12,743 • GYKI-16,084 • Idazoxan • Mianserin • Mirtazapine • MK-912 • NAN-190 • Olanzapine • Phentolamine • Phenoxybenzamine • Piperoxan • Piribedil • Rauwolscine • Rotigotine • SB-269,970 • Setiptiline • Spiroxatrine • Sunepitron • Tolazoline • Yohimbine
* Note that many atypical antipsychotics and azapirones like buspirone and gepirone (via metabolite 1-PP) antagonize α2-adrenergic receptors as well.βAgonists: 2-FNE • 5-FNE • Amibegron • Arbutamine • Arformoterol • Arotinolol • BAAM • Bambuterol • Befunolol • Bitolterol • Broxaterol • Buphenine • Carbuterol • Cimaterol • Clenbuterol • Denopamine • Deterenol • Dipivefrine • Dobutamine • Dopamine • Dopexamine • Ephedrine • Epinephrine (Adrenaline) • Etafedrine • Etilefrine • Ethylnorepinephrine • Fenoterol • Formoterol • Hexoprenaline • Higenamine • Indacaterol • Isoetarine • Isoprenaline (Isoproterenol) • Isoxsuprine • Labetalol • Levonordefrin • Levosalbutamol • Mabuterol • Methoxyphenamine • Methyldopa • Norepinephrine (Noradrenaline) • Orciprenaline • Oxyfedrine • Phenylpropanolamine • Pirbuterol • Prenalterol • Ractopamine • Procaterol • Pseudoephedrine • Reproterol • Rimiterol • Ritodrine • Salbutamol (Albuterol) • Salmeterol • Solabegron • Terbutaline • Tretoquinol • Tulobuterol • Xamoterol • Zilpaterol • Zinterol
Antagonists: Acebutolol • Adaprolol • Adimolol • Afurolol • Alprenolol • Alprenoxime • Amosulalol • Ancarolol • Arnolol • Arotinolol • Atenolol • Befunolol • Betaxolol • Bevantolol • Bisoprolol • Bopindolol • Bormetolol • Bornaprolol • Brefonalol • Bucindolol • Bucumolol • Bufetolol • Buftiralol • Bufuralol • Bunitrolol • Bunolol • Bupranolol • Burocrolol • Butaxamine • Butidrine • Butofilolol • Capsinolol • Carazolol • Carpindolol • Carteolol • Carvedilol • Celiprolol • Cetamolol • Cicloprolol • Cinamolol • Cloranolol • Cyanopindolol • Dalbraminol • Dexpropranolol • Diacetolol • Dichloroisoprenaline • Dihydroalprenolol • Dilevalol • Diprafenone • Draquinolol • Dropranolol • Ecastolol • Epanolol • Ericolol • Ersentilide • Esatenolol • Esmolol • Esprolol • Eugenodilol • Exaprolol • Falintolol • Flestolol • Flusoxolol • Hydroxycarteolol • Hydroxytertatolol • ICI-118,551 • Idropranolol • Indenolol • Indopanolol • Iodocyanopindolol • Iprocrolol • Isoxaprolol • Isamoltane • Labetalol • Landiolol • Levobetaxolol • Levobunolol • Levocicloprolol • Levomoprolol • Medroxalol • Mepindolol • Metalol • Metipranolol • Metoprolol • Moprolol • Nadolol • Nadoxolol • Nafetolol • Nebivolol • Neraminol • Nifenalol • Nipradilol • Oberadilol • Oxprenolol • Pacrinolol • Pafenolol • Pamatolol • Pargolol • Parodilol • Penbutolol • Penirolol • PhQA-33 • Pindolol • Pirepolol • Practolol • Primidolol • Procinolol • Pronethalol • Propafenone • Propranolol • Ridazolol • Ronactolol • Soquinolol • Sotalol • Spirendolol • SR 59230A • Sulfinalol • TA-2005 • Talinolol • Tazolol • Teoprolol • Tertatolol • Terthianolol • Tienoxolol • Tilisolol • Timolol • Tiprenolol • Tolamolol • Toliprolol • Tribendilol • Trigevolol • Xibenolol • XipranololReuptake inhibitors Selective norepinephrine reuptake inhibitors: Amedalin • Atomoxetine (Tomoxetine) • Ciclazindol • Daledalin • Esreboxetine • Lortalamine • Mazindol • Nisoxetine • Reboxetine • Talopram • Talsupram • Tandamine • Viloxazine; Norepinephrine-dopamine reuptake inhibitors: Amineptine • Bupropion (Amfebutamone) • Fencamine • Fencamfamine • Lefetamine • Levophacetoperane • LR-5182 • Manifaxine • Methylphenidate • Nomifensine • O-2172 • Radafaxine; Serotonin-norepinephrine reuptake inhibitors: Bicifadine • Desvenlafaxine • Duloxetine • Eclanamine • Levomilnacipran • Milnacipran • Sibutramine • Venlafaxine; Serotonin-norepinephrine-dopamine reuptake inhibitors: Brasofensine • Diclofensine • DOV-102,677 • DOV-21,947 • DOV-216,303 • JNJ-7925476 • JZ-IV-10 • Methylnaphthidate • Naphyrone • NS-2359 • PRC200-SS • SEP-225,289 • SEP-227,162 • Tesofensine; Tricyclic antidepressants: Amitriptyline • Butriptyline • Cianopramine • Clomipramine • Desipramine • Dosulepin • Doxepin • Imipramine • Lofepramine • melitracen • Nortriptyline • Protriptyline • Trimipramine; Tetracyclic antidepressants: Amoxapine • Maprotiline • Mianserin • Oxaprotiline • Setiptiline; Others: Cocaine • CP-39,332 • EXP-561 • Fezolamine • Ginkgo biloba • Indeloxazine • Nefazodone • Nefopam • Pridefrine • Tapentadol • Teniloxazine • Tramadol • ZiprasidoneEnzyme inhibitors 3,4-DihydroxystyreneDBHCGS-19281A • SKF-64139 • SKF-7698Nonselective: Benmoxin • Caroxazone • Echinopsidine • Furazolidone • Hydralazine • Indantadol • Iproclozide • Iproniazid • Isocarboxazid • Isoniazid • Linezolid • Mebanazine • Metfendrazine • Nialamide • Octamoxin • Paraxazone • Phenelzine • Pheniprazine • Phenoxypropazine • Pivalylbenzhydrazine • Procarbazine • Safrazine • Tranylcypromine; MAO-A selective: Amiflamine • Bazinaprine • Befloxatone • Befol • Brofaromine • Cimoxatone • Clorgiline • Esuprone • Harmala alkaloids (Harmine, Harmaline, Tetrahydroharmine, Harman, Norharman, etc) • Methylene Blue • Metralindole • Minaprine • Moclobemide • Pirlindole • Sercloremine • Tetrindole • Toloxatone • Tyrima; MAO-B selective: D-Deprenyl • Selegiline (L-Deprenyl) • Ladostigil • Lazabemide • Milacemide • Mofegiline • Pargyline • Rasagiline • Safinamide
* Note that MAO-B inhibitors also influence norepinephrine/epinephrine levels since they inhibit the breakdown of their precursor dopamine.COMTOthers Ferrous Iron (Fe2+) • S-Adenosyl-L-Methionine • Vitamin B3 (Niacin, Nicotinamide → NADPH) • Vitamin B6 (Pyridoxine, Pyridoxamine, Pyridoxal → Pyridoxal Phosphate) • Vitamin B9 (Folic acid → Tetrahydrofolic acid) • Vitamin C (Ascorbic acid) • Zinc (Zn2+)OthersActivity enhancers: BPAP • PPAP; Release blockers: Bethanidine • Bretylium • Guanadrel • Guanazodine • Guanclofine • Guanethidine • Guanoxan; Toxins: Oxidopamine (6-Hydroxydopamine)List of adrenergic drugsPhenethylamines Phenethylamines Psychedelics: 2C-B • 2C-B-FLY • 2C-C • 2C-D • 2C-E • 2C-F • 2C-G • 2C-I • 2C-N • 2C-P • 2C-SE • 2C-T • 2C-T-2 • 2C-T-4 • 2C-T-7 • 2C-T-8 • 2C-T-9 • 2C-T-13 • 2C-T-15 • 2C-T-17 • 2C-T-21 • 2C-TFM • 2C-YN • Allylescaline • DESOXY • Escaline • Isoproscaline • Jimscaline • Macromerine • MEPEA • Mescaline • Metaescaline • Methallylescaline • Proscaline • Psi-2C-T-4 • TCB-2
Stimulants: 2-OH-PEA • β-Me-PEA • Hordenine • N-Me-PEA • Phenethylamine (PEA)
Entactogens: Lophophine • MDPEA • MDMPEA
Others: BOH • DMPEAAmphetamines
PhenylisopropylaminesPsychedelics: 3C-BZ • 3C-E • 3C-P • Aleph • Beatrice • Bromo-DragonFLY • D-Deprenyl • DMA • DMCPA • DMMDA • DOB • DOC • DOEF • DOET • DOI • DOM • DON • DOPR • DOTFM • Ganesha • MMDA • MMDA-2 • Psi-DOM • TMA • TeMA
Stimulants: 4-MA • 4-MMA • 4-MTA • 5-IT • Alfetamine • Amfecloral • Amfepentorex • Amphetamine (Dextroamphetamine, Levoamphetamine) • Amphetaminil • Benfluorex • Benzphetamine • Cathine • Clobenzorex • Dimethylamphetamine • Ephedrine (EPH) • Ethylamphetamine • Fencamfamine • Fencamine • Fenethylline • Fenfluramine (Dexfenfluramine) • Fenproporex • Fludorex • Furfenorex • Isopropylamphetamine • Lefetamine • Mefenorex • Methamphetamine (Dextromethamphetamine, Levomethamphetamine) • Methoxyphenamine • MMA • Norfenfluramine • Oxilofrine • Ortetamine • PBA • PCA • Phenpromethamine • PFA • PFMA • PIA • PMA • PMEA • PMMA • Phenylpropanolamine (PPA) • Prenylamine • Propylamphetamine • Pseudoephedrine (PSE) • Sibutramine • Tiflorex (Flutiorex) • Tranylcypromine • Xylopropamine • Zylofuramine
Entactogens: 5-APDB • 6-APB • 6-APDB • EDA • IAP • MDA • MDEA • MDHMA (FLEA) • MDMA ("Ecstasy") • MDOH • MMDMA • NAP • TAP
Others: Amiflamine • DFMDA • D-Deprenyl • L-Deprenyl (Selegiline)Phentermines Stimulants: Chlorphentermine • Cloforex • Clortermine • Etolorex • Mephentermine • Pentorex (Phenpentermine) • Phentermine
Entactogens: MDPH • MDMPHCathinones Stimulants: Amfepramone • Brephedrone • Buphedrone • Bupropion (Amfebutamone) • Cathinone (Propion) • Dimethylcathinone (Dimethylpropion, Metamfepramone) • Ethcathinone (Ethylpropion) • Flephedrone • Methcathinone (Methylpropion) • Mephedrone • Methedrone
Entactogens: Ethylone • MethylonePhenylisobutylamines Phenylalkylpyrrolidines Stimulants: α-PBP • α-PPP • α-PVP • MDPBP • MDPPP • MDPV • MOPPP • MPBP • MPHP • MPPP • Naphyrone • PEP • Prolintane • PyrovaleroneCatecholamines
(and relatives..)6-FNE • 6-OHDA • α-Me-DA • α-Me-TRA • Adrenochrome • Ciladopa • D-DOPA (Dextrodopa) • Dopamine • Epinephrine (Adrenaline) • Epinine • Fenclonine • Ibopamine • L-DOPA (Levodopa) • L-DOPS (Droxidopa) • L-Phenylalanine • L-Tyrosine • meta-Octopamine • meta-Tyramine • Metanephrine • Metirosine • Methyldopa • Nordefrin (Levonordefrin) • Norepinephrine (Noradrenaline) • Normetanephrine • para-Octopamine • para-TyramineMiscellaneous Amidephrine • Arbutamine • Cafedrine • Denopamine • Dobutamine • Dopexamine • Etafedrine • Ethylnorepinephrine • Etilefrine • Famprofazone • Gepefrine • Isoprenaline (Isoproterenol) • Isoetarine • Metaraminol • Metaterol • Methoxamine • Norfenefrine • Orciprenaline • Phenylephrine (Neosynephrine) • Phenoxybenzamine • Prenalterol • Pronethalol • Propranolol • Salbutamol (Albuterol; Levosalbutamol) • Synephrine (Oxedrine) • Theodrenaline • XamoterolCategories:- Amphetamine alkaloids
- Decongestants
- World Health Organization essential medicines
Wikimedia Foundation. 2010.