- Penicillamine
-
Penicillamine 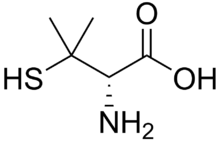
Systematic (IUPAC) name (2S)-2-amino-3-methyl-3-sulfanyl-butanoic acid Clinical data Trade names Cuprimine AHFS/Drugs.com monograph Pregnancy cat. D (Aust.) Legal status Prescription Routes Oral Pharmacokinetic data Bioavailability Variable Metabolism Hepatic Half-life 1 hour Excretion Renal Identifiers CAS number 52-67-5 
ATC code M01CC01 PubChem CID 5852 DrugBank APRD01171 ChemSpider 5643 
UNII GNN1DV99GX 
KEGG D00496 
ChEBI CHEBI:7959 
ChEMBL CHEMBL1430 
Chemical data Formula C5H11NO2S Mol. mass 149.212 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Penicillamine is a pharmaceutical of the chelator class. It is sold under the trade names of Cuprimine and Depen. The pharmaceutical form is D-penicillamine, as L-penicillamine is toxic (it inhibits the action of pyridoxine). It is a metabolite of penicillin, although it has no antibiotic properties.
Contents
Uses
Penicillamine is used as a form of immunosuppression to treat rheumatoid arthritis. It works by reducing numbers of T-lymphocytes, inhibiting macrophage function, decreasing IL-1, decreasing rheumatoid factor, and preventing collagen from cross-linking.
It is used as a chelating agent:
- In Wilson's disease, a rare genetic disorder of copper metabolism, penicillamine treatment relies on its binding to accumulated copper and elimination through urine.
- In cystinuria, a hereditary disorder featuring formation of cystine stones, penicillamine binds with cysteine to yield a mixed disulfide which is more soluble than cystine.
- Penicillamine has been used to treat scleroderma
- Penicillamine is the second line treatment for arsenic poisoning, after dimercaprol (BAL)
Adverse effects
Adverse effects include:
- Membranous glomerulonephritis [1]
- Aplastic anemia (idiosyncratic) [2]
- Antibody-mediated myasthenic syndrome, which may persist even after its withdrawal
- Drug-induced systemic lupus erythematosus
- Elastosis perforans serpiginosa [3]
- Toxic myopathies[4]
History
Dr. John Walshe (1956) first described the use of penicillamine in Wilson's disease.[5] He had discovered the compound in the urine of patients (including himself) who had taken penicillin, and experimentally confirmed that it increased urinary copper excretion by chelation. He had initial difficulty convincing several world experts of the time (Drs Denny Brown and Cumings) of its efficacy, as they held that Wilson's disease was not primarily a problem of copper homeostasis but of amino acid metabolism, and that dimercaprol should be used as a chelator. Later studies confirmed both the copper-centered theory and the efficacy of D-penicillamine. Walshe also pioneered other chelators in Wilson's such as triethylene tetramine, 2HCl, and tetrathiomolybdate.[6]
References
- ^ Table 14-2 in: Mitchell, Richard Sheppard; Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson. Robbins Basic Pathology. Philadelphia: Saunders. ISBN 1-4160-2973-7. 8th edition.
- ^ Robbins and Cotran, Pathological Basis of Disease 8th Edition, Kumar et al
- ^ Bolognia, Jean; et al (2007). Dermatology. Philadelphia: Elsevier. ISBN 1416029990.2nd edition.
- ^ Underwood, J. C. E. (2009). General and systemic Pathology. Elsevier Limited. ISBN 9780443068898.
- ^ Walshe JM (January 1956). "Wilson's disease; new oral therapy". Lancet 267 (6906): 25–6. doi:10.1016/S0140-6736(56)91859-1. PMID 13279157.
- ^ Walshe JM (August 2003). "The story of penicillamine: a difficult birth". Mov. Disord. 18 (8): 853–9. doi:10.1002/mds.10458. PMID 12889074.
External links
- Penicillamine (Systemic) - Medlineplus.org
- Penicillamine and Arthritis - Medicinenet.com
Specific antirheumatic products / DMARDs (M01C) Quinolines Gold preparations Other Penicillamine #/Bucillamine • Chloroquine #/Hydroxychloroquine • Leflunomide • Sulfasalazine # • antifolate (Methotrexate #) • thiopurine (Azathioprine) #M: JNT
anat(h/c, u, t, l)/phys
noco(arth/defr/back/soft)/cong, sysi/epon, injr
proc, drug(M01C, M4)
Chelating agents / chelation therapy (V03AC, others) Iron Copper Penicillamine#Lead Thallium Other/ungrouped Categories:- Enantiopure drugs
- Amino acids
- Chelating agents
- Antirheumatic products
- Thiols
- World Health Organization essential medicines
Wikimedia Foundation. 2010.
